Pricing and Reimbursement of Biosimilars in Central and Eastern European Countries
- PMID: 28642700
- PMCID: PMC5463127
- DOI: 10.3389/fphar.2017.00288
Pricing and Reimbursement of Biosimilars in Central and Eastern European Countries
Abstract
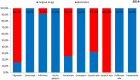
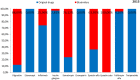
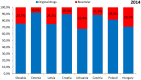
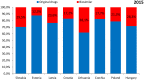
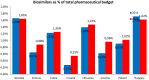
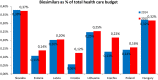
Objectives: The aim of this study was to review the requirements for the reimbursement of biosimilars and to compare the reimbursement status, market share, and reimbursement costs of biosimilars in selected Central and Eastern European (CEE) countries. Methods: A questionnaire-based survey was conducted between November 2016 and January 2017 among experts from the following CEE countries: Bulgaria, Czech Republic, Croatia, Estonia, Hungary, Latvia, Lithuania, Poland, Slovakia, and Romania. The requirements for the pricing and reimbursement of biosimilars were reviewed for each country. Data on the extent of reimbursement of biologic drugs (separately for original products and biosimilars) in the years 2014 and 2015 were also collected for each country, along with data on the total pharmaceutical and total public health care budgets. Results: Our survey revealed that no specific criteria were applied for the pricing and reimbursement of biosimilars in the selected CEE countries; the price of biosimilars was usually reduced compared with original drugs and specific price discounts were common. Substitution and interchangeability were generally allowed, although in most countries they were at the discretion of the physician after a clinical assessment. Original biologic drugs and the corresponding biosimilars were usually in the same homogeneous group, and internal reference pricing was usually employed. The reimbursement rate of biosimilars in the majority of the countries was the same and amounted to 100%. Generally, the higher shares of expenditures were shown for the reimbursement of original drugs than for biosimilars, except for filgrastim, somatropin, and epoetin (alfa and zeta). The shares of expenditures on the reimbursement of biosimilar products ranged from 8.0% in Estonia in 2014 to 32.4% in Lithuania in 2015, and generally increased in 2015. The share of expenditures on reimbursement of biosimilars in the total pharmaceutical budget differed between the countries, with the highest observed value for Slovakia and Hungary and the lowest-for Croatia. Conclusions: The requirements for the pricing and reimbursement of biosimilar products as well as the access of patients to biologic treatment do not differ significantly between the considered CEE countries. Biosimilar drugs significantly influence the reimbursement systems of these countries, and the expenditure on the reimbursement of biosimilars is increasing as they are becoming more accessible to patients.
Keywords: biosimilar pharmaceuticals; interchangeability; original products; pricing; reimbursement.
Figures
References
-
- Brodszky V., Rencz F., Péntek M., Baji P., Lakatos P. L., Gulácsi L. (2016). A budget impact model for biosimilar infliximab in Crohn's disease in Bulgaria, the Czech Republic, Hungary, Poland, Romania, and Slovakia. Expert Rev. Pharmacoecon. Outcomes Res. 16, 119–125. 10.1586/14737167.2015.1067142 - DOI - PubMed
-
- Derbyshire M. (2015). Patent expiry dates for best-selling biological. Gene. Biosimilars Initiat. J. 4, 178–179. 10.5639/gabij.2015.0404.040 - DOI
-
- ESzCsM Decree (2017). 32/2004. (IV. 26.) ESzCsM Decree, Concerning the Reimbursement of Medicinal Products. Available online at: http://net.jogtar.hu/jr/gen/hjegy_doc.cgi?docid=A0400032.ESC (Accessed February 13, 2017).
-
- European Medicines Agency (2017). European Public Assessment Reports. Available online at: http://www.ema.europa.eu/ema/index.jsp?curl=pages%2Fmedicines%2Flanding%... (Accessed February 13, 2017).
LinkOut - more resources
Full Text Sources
Other Literature Sources

