The number of injected same-day preschool vaccines relates to preadolescent needle fear and HPV uptake
- PMID: 28647169
- PMCID: PMC5570478
- DOI: 10.1016/j.vaccine.2017.06.029
The number of injected same-day preschool vaccines relates to preadolescent needle fear and HPV uptake
Abstract
Purpose: Fear of needles develops at approximately five years of age, and decreases compliance with healthcare. We sought to examine the relationship of preschool vaccine history, parent and preadolescent needle fear, and subsequent compliance with optional vaccines.
Methods: As part of a private practice randomized controlled trial, parents and 10-12year olds rated needle anxiety on a 100mm visual analog scale. This follow-up cohort study compared their needle anxiety to previous vaccination records, including number of vaccinations between ages four and six years (total and same-day maximum), and subsequent initiation of the HPV vaccine through age 13.
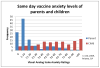
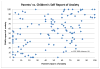
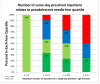
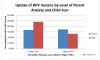
Results: Of the 120 preadolescents enrolled between 4.28.09 and 1.19.2010, 117 received preschool vaccinations between ages four and six years. The likelihood of being in the upper quartile of fear (VAS≥83) five years later increased with each additional same-day injection (OR=3.108, p=0.0100 95%CI=1.311, 7.367), but was not related to total lifetime or total four-to-six year injections. Only 12.5% (15) of parents reported anxiety about their preadolescents' vaccines (VAS>50). Parent and child anxiety was weakly correlated (r=0.15). Eight children in the upper fear quartile began their HPV series (26.67%) compared to 14 in the lower quartile (48.28% VAS<32) (OR 2.57, p=0.0889, 95%CI 0.864-7.621); there was no difference in HPV uptake between upper and lower quartile of parent anxiety.
Conclusions: The more same-day preschool injections between 4 and 6years of age, the more likely a child was to fear needles five years later. Preadolescent needle fear was a stronger predictor than parent vaccine anxiety of subsequent HPV vaccine uptake.
Keywords: Adherence; HPV; Needle fear; Pain; Vaccine Hesitancy; phobia.
Copyright © 2017 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Conflict of Interest: Dr. Baxter invented Buzzy and is the CEO of MMJ Labs, the manufacturer of Buzzy®. This conflict of interest was disclosed to participants in the informed consent. After orientation of enrollers employed by the hospital, Dr. Baxter was not present for data collection. Dr. Cohen, who has no financial conflict of interest to disclose, recruited study coordinators.
Declaration of Interests
The initial NIH grant supported research and development of a needle pain device created by Amy Baxter MD. Dr. Baxter created a company to manufacture and market the device, and has subsequent conflict of interest; this study does not refer to the device. Lindsey Cohen PhD, M. Louise Lawson PhD, and Mark Burton have no conflicts of interest. Anaam Mohammed worked as a study coordinator for Dr. Baxter when she ran a pediatric emergency research division but otherwise has no conflict of interest. All funding for the study was provided by the NIH grant 4R44HD056647-02.
Figures
References
-
- Froehlich H, West DJ. Compliance with hepatitis B virus vaccination in a high-risk population. Ethn Dis. 2001;11:548–53. - PubMed
-
- Meyerhoff AS, Weniger BG, Jacobs RJ. Economic value to parents of reducing the pain and emotional distress of childhood vaccine injections. Pediatr Infect Dis J. 2001;20:S57–62. - PubMed
-
- Taddio A, Ipp M, Thivakaran S, et al. Survey of the prevalence of immunization non-compliance due to needle fears in children and adults. Vaccine. 2012;30:4807–12. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

