Isolation and structural characterization of dihydrobenzofuran congeners of licochalcone A
- PMID: 28647482
- PMCID: PMC5660606
- DOI: 10.1016/j.fitote.2017.06.017
Isolation and structural characterization of dihydrobenzofuran congeners of licochalcone A
Abstract
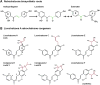
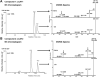
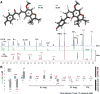
In an effort to explore the residual complexity of naturally occurring chalcones from the roots of Glycyrrhiza inflata (Fabaceae), two new licochalcone A (LicA) derivatives were isolated as trace metabolites from enriched fractions. Both constituents contain a dihydrofuran moiety linked to carbons C-4 and C-5 of the retrochalcone core. Compound 1 (LicAF1) represents a new chemical entity, whereas compound 2 (LicAF2) has previously been reported as a Lewis acid catalyzed rearrangement of LicA. Evaluation of chirality revealed that both dihydrofuran derivatives existed as a mixture of R and S enantiomers. Interestingly, when solutions were exposed to sunlight, both dihydrofuran retrochalcones, initially isolated as trans isomers, were found to rapidly isomerize yielding trans and cis isomers. Analysis of the 1D 1H NMR spectra of the photolysis products revealed the presence of two sets of proton resonances ascribed to each of the geometric isomers. An up-field shift of all proton resonances arising from the cis isomer was observed, suggesting that anisotropic shielding effects were introduced through an overall perturbation of the 3-dimensional structure upon photoisomerization. Similar up-field shifts were observed in the 13C spectrum of the cis isomer, except for the CO, C-α, and C-6 carbons, which experienced downfield shifts. Analogous NMR results were observed for LicA. Hence, the results presented herein encompass the isolation and full characterization of LicAF analogs 1 and 2, and observations of their trans-to-cis photoisomerization through the systematic analysis of their NMR spectra.
Keywords: Cis/trans isomerization; Dihydrobenzofuran; Licochalcone A; Raw NMR data; Residual complexity; Retrochalcone.
Copyright © 2017 Elsevier B.V. All rights reserved.
Conflict of interest statement
Figures



References
-
- Shibata S, et al. A drug over the millennia: pharmacognosy, chemistry, and pharmacology of licorice. Yakugaku Zasshi J Pharm Soc Japan. 2000;120:849. - PubMed
-
- Chen KZ, Song H, Ruyu C. Licorice industry in China∷ Implications for licorice producers in Uzbekistan. 2014
-
- Farag MA, Porzel A, Wessjohann LA. Comparative metabolite profiling and fingerprinting of medicinal licorice roots using a multiplex approach of GC-MS, LC-MS and 1D NMR techniques. Phytochemistry. 2012;76:60–72. - PubMed
-
- Kajiyama K, Demizu S, Hiraga Y, Kinoshita K, Takahashi K. Two Prenylated Retrochalcones from Glycyrrhiza inflata. Phytochemistry. 1992;31:3229–3232.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

