Aryl hydrocarbon receptor (AHR) is a potential tumour suppressor in pituitary adenomas
- PMID: 28649092
- PMCID: PMC5541251
- DOI: 10.1530/ERC-17-0112
Aryl hydrocarbon receptor (AHR) is a potential tumour suppressor in pituitary adenomas
Abstract
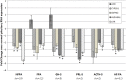
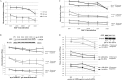
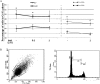
Pituitary adenomas (PA) represent the largest group of intracranial neoplasms and yet the molecular mechanisms driving this disease remain largely unknown. The aim of this study was to use a high-throughput screening method to identify molecular pathways that may be playing a significant and consistent role in PA. RNA profiling using microarrays on eight local PAs identified the aryl hydrocarbon receptor (AHR) signalling pathway as a key canonical pathway downregulated in all PA types. This was confirmed by real-time PCR in 31 tumours. The AHR has been shown to regulate cell cycle progression in various cell types; however, its role in pituitary tissue has never been investigated. In order to validate the role of AHR in PA behaviour, further functional studies were undertaken. Over-expression of AHR in GH3 cells revealed a tumour suppressor potential independent of exogenous ligand activation by benzo α-pyrene (BαP). Cell cycle analysis and quantitative PCR of cell cycle regulator genes revealed that both unstimulated and BαP-stimulated AHR reduced E2F-driven transcription and altered expression of cell cycle regulator genes, thus increasing the percentage of cells in G0/G1 phase and slowing the proliferation rate of GH3 cells. Co-immunoprecipitation confirmed the interaction between AHR and retinoblastoma (Rb1) protein supporting this as a functional mechanism for the observed reduction. Endogenous Ahr reduction using silencing RNA confirmed the tumour suppressive function of the Ahr. These data support a mechanistic pathway for the putative tumour suppressive role of AHR specifically in PA, possibly through its role as a cell cycle co-regulator, even in the absence of exogenous ligands.
Keywords: AHR; microarray; pituitary adenoma; tumour suppressor.
© 2017 The authors.
Figures


References
-
- Asa S, Casar-Borota O, Chanson P, Delgrange E, Earls P, Ezzat S, Grossman A, Ikeda H, Inoshita N, Karavitaki N, et al. 2017. From pituitary adenoma to pituitary neuroendocrine tumour (PitNET): an International Pituitary Pathology Club proposal. Endocrine-Related Cancer 24 C5–C8. ( 10.1530/ERC-17-0004) - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

