Organ sculpting by patterned extracellular matrix stiffness
- PMID: 28653906
- PMCID: PMC5503509
- DOI: 10.7554/eLife.24958
Organ sculpting by patterned extracellular matrix stiffness
Abstract
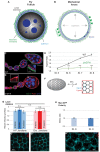
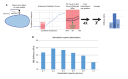
How organ-shaping mechanical imbalances are generated is a central question of morphogenesis, with existing paradigms focusing on asymmetric force generation within cells. We show here that organs can be sculpted instead by patterning anisotropic resistance within their extracellular matrix (ECM). Using direct biophysical measurements of elongating Drosophila egg chambers, we document robust mechanical anisotropy in the ECM-based basement membrane (BM) but not in the underlying epithelium. Atomic force microscopy (AFM) on wild-type BM in vivo reveals an anterior-posterior (A-P) symmetric stiffness gradient, which fails to develop in elongation-defective mutants. Genetic manipulation shows that the BM is instructive for tissue elongation and the determinant is relative rather than absolute stiffness, creating differential resistance to isotropic tissue expansion. The stiffness gradient requires morphogen-like signaling to regulate BM incorporation, as well as planar-polarized organization to homogenize it circumferentially. Our results demonstrate how fine mechanical patterning in the ECM can guide cells to shape an organ.
Keywords: D. melanogaster; atomic force microscopy; basement membrane; cell biology; developmental biology; extracellular matrix; morphogenesis; stem cells.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures










Comment in
-
Morphogenesis: Shaping Tissues through Extracellular Force Gradients.Curr Biol. 2017 Sep 11;27(17):R850-R852. doi: 10.1016/j.cub.2017.07.040. Curr Biol. 2017. PMID: 28898649
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

