Evaluation of early antimicrobial therapy adaptation guided by the BetaLACTA® test: a case-control study
- PMID: 28655352
- PMCID: PMC5488410
- DOI: 10.1186/s13054-017-1746-6
Evaluation of early antimicrobial therapy adaptation guided by the BetaLACTA® test: a case-control study
Abstract
Background: Rapid diagnostic tests detecting microbial resistance are needed for limiting the duration of inappropriateness of empirical antimicrobial therapy (EAT) in intensive care unit patients, besides reducing the use of broad-spectrum antibiotics. We hypothesized that the betaLACTA® test (BLT) could lead to early increase in the adequacy of antimicrobial therapy.
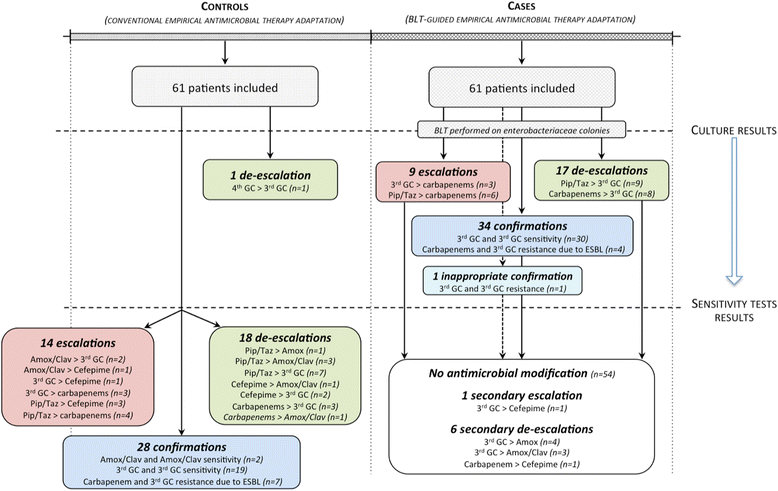
Methods: This was a case-control study. Sixty-one patients with BLT-guided adaptation of EAT were prospectively included, and then matched with 61 "controls" having similar infection characteristics (community or hospital-acquired, and source of infection), in whom EAT was conventionally adapted to antibiogram results. Endpoints were to compare the proportion of appropriate (primary endpoint) and optimal (secondary endpoint) antimicrobial therapies with each of the two strategies, once microbiological sample culture results were available.
Results: Characteristics of patients, infections and EAT at inclusion were similar between groups. Nine early escalations of EAT occurred in the BLT-guided adaptation group, reaching 98% appropriateness vs. 77% in the conventional adaptation group (p < 0.01). The BLT reduced the time until escalation of an inappropriate EAT from 50.5 (48-73) to 27 (24-28) hours (p < 0.01). Seventeen early de-escalations occurred in the BLT-guided adaptation group, compared to one in the conventional adaptation group, reducing patients' exposure to broad-spectrum beta-lactam such as carbapenems. In multivariate analysis, use of the BLT was strongly associated with early appropriate (OR = 18 (3.4-333.8), p = 0.006) and optimal (OR = 35.5 (9.6-231.9), p < 0.001) antimicrobial therapies. Safety parameters were similar between groups.
Conclusions: Our study suggests that a BLT-guided adaptation strategy may allow early beta-lactam adaptation from the first 24 hours following the beginning of sepsis management.
Keywords: Antimicrobial agent administration; Beta-lactam resistance; Extended-spectrum beta-lactamase; Intensive care unit; Microbial susceptibility tests.
Figures


Comment in
-
Rapid identification of antimicrobial resistance patterns allows a faster antibiotic adequacy.Crit Care. 2017 Aug 4;21(1):208. doi: 10.1186/s13054-017-1797-8. Crit Care. 2017. PMID: 28784143 Free PMC article. No abstract available.
References
-
- InVS/REA-Raisin. Surveillance de la consommation des antibiotiques (Antimicrobials consumption monitoring) (2011) [Internet]. 2013. Available from: http://www.invs.sante.fr/Publications-et-outils/Rapports-et-syntheses/Ma.... 14 June 2017.
-
- European Centre for Disease Prevention and Control. Antimicrobial resistance interactive database (2013) [Internet]. 2013. Available from: http://www.ecdc.europa.eu/en/healthtopics/antimicrobial_resistance/datab.... 15 Apr 2017.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

