A genetic risk score is differentially associated with migraine with and without aura
- PMID: 28656458
- PMCID: PMC5502071
- DOI: 10.1007/s00439-017-1816-5
A genetic risk score is differentially associated with migraine with and without aura
Abstract
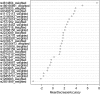
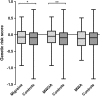
Although a number of migraine-associated single-nucleotide polymorphisms (SNP) with small effect size have been identified, little is known about the additive impact of these variants on migraine risk, frequency and severity. We investigated to what extent a genetic risk score (GRS) based on recently published, novel migraine-associated SNPs is associated with migraine prevalence, subtypes and severity in a large population-based sample. The sample comprised 446 subjects with migraine and 2511 controls from the CoLaus/PsyCoLaus study. Fifty-four SNPs earlier associated with migraine were selected. SNPs with a low impact on migraine prevalence in our sample were excluded using random forest. We combined the remaining 21 SNPs into a GRS and analyzed the association with migraine using logistic regression models. The GRS was significantly associated with migraine (OR = 1.56, p = 0.02) and migraine without aura (MWOA) (OR = 2.01, p = 0.003), but not with migraine with aura (MWA). The GRS was not associated with migraine frequency, intensity or interference with daily activities. We show that a GRS combining multiple genetic risk variants is associated with MWOA but not MWA, suggesting a different genetic susceptibility background underlying the two forms of migraine.
Conflict of interest statement
Funding
This study was supported by a grant from the Swedish Society for Medical Research (SSMF) to JM, a grant from the Swedish Research Council to HBS, and in part by the Intramural Research Program of the National Institute of Mental Health (ZIAMH002932). The CoLaus|PsyCoLaus study was and is supported by research grants from GlaxoSmithKline, the Faculty of Biology and Medicine of Lausanne, the Swiss National Science Foundation (Grants 3200B0-105993, 3200B0-118308, 33CSCO-122661, 33CS30-139468, and 33CS30-148401).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Figures
References
-
- Bettschart W, Bolognini M. Questionnaire de santé GHQ-12. Boulogne: Médicales Pierre Fabre; 1996.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

