Activin receptor type 2A (ACVR2A) functions directly in osteoblasts as a negative regulator of bone mass
- PMID: 28659341
- PMCID: PMC5566533
- DOI: 10.1074/jbc.M117.782128
Activin receptor type 2A (ACVR2A) functions directly in osteoblasts as a negative regulator of bone mass
Abstract
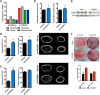
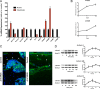
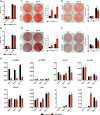
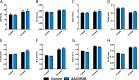
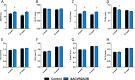
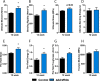
Bone and skeletal muscle mass are highly correlated in mammals, suggesting the existence of common anabolic signaling networks that coordinate the development of these two anatomically adjacent tissues. The activin signaling pathway is an attractive candidate to fulfill such a role. Here, we generated mice with conditional deletion of activin receptor (ACVR) type 2A, ACVR2B, or both, in osteoblasts, to determine the contribution of activin receptor signaling in regulating bone mass. Immunohistochemistry localized ACVR2A and ACVR2B to osteoblasts and osteocytes. Primary osteoblasts expressed activin signaling components, including ACVR2A, ACVR2B, and ACVR1B (ALK4) and demonstrated increased levels of phosphorylated Smad2/3 upon exposure to activin ligands. Osteoblasts lacking ACVR2B did not show significant changes in vitro However, osteoblasts deficient in ACVR2A exhibited enhanced differentiation indicated by alkaline phosphatase activity, mineral deposition, and transcriptional expression of osterix, osteocalcin, and dentin matrix acidic phosphoprotein 1. To investigate activin signaling in osteoblasts in vivo, we analyzed the skeletal phenotypes of mice lacking these receptors in osteoblasts and osteocytes (osteocalcin-Cre). Similar to the lack of effect in vitro, ACVR2B-deficient mice demonstrated no significant change in any bone parameter. By contrast, mice lacking ACVR2A had significantly increased femoral trabecular bone volume at 6 weeks of age. Moreover, mutant mice lacking both ACVR2A and ACVR2B demonstrated sustained increases in trabecular bone volume, similar to those in ACVR2A single mutants, at 6 and 12 weeks of age. Taken together, these results indicate that activin receptor signaling, predominantly through ACVR2A, directly and negatively regulates bone mass in osteoblasts.
Keywords: activin; bone; myostatin; osteoblast; transforming growth factor beta (TGF-β).
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures


Similar articles
-
BMP3 suppresses osteoblast differentiation of bone marrow stromal cells via interaction with Acvr2b.Mol Endocrinol. 2012 Jan;26(1):87-94. doi: 10.1210/me.2011-1168. Epub 2011 Nov 10. Mol Endocrinol. 2012. PMID: 22074949 Free PMC article.
-
Murine FSH Production Depends on the Activin Type II Receptors ACVR2A and ACVR2B.Endocrinology. 2020 Jul 1;161(7):bqaa056. doi: 10.1210/endocr/bqaa056. Endocrinology. 2020. PMID: 32270195 Free PMC article.
-
Loss of BMPR2 leads to high bone mass due to increased osteoblast activity.J Cell Sci. 2015 Apr 1;128(7):1308-15. doi: 10.1242/jcs.156737. Epub 2015 Feb 6. J Cell Sci. 2015. PMID: 25663702 Free PMC article.
-
Targeting osteocytes vs osteoblasts.Bone. 2023 May;170:116724. doi: 10.1016/j.bone.2023.116724. Epub 2023 Mar 1. Bone. 2023. PMID: 36868508 Free PMC article. Review.
-
Bioinformatics Analysis of the Genetic and Epigenetic Alterations of Bone Morphogenetic Protein Receptors in Metastatic Breast Cancer.Biochem Genet. 2024 Apr;62(2):594-620. doi: 10.1007/s10528-023-10445-2. Epub 2023 Jul 24. Biochem Genet. 2024. PMID: 37486509 Review.
Cited by
-
Deciphering the Functional Roles of Individual Cancer Alleles Across Comprehensive Cancer Genomic Studies.bioRxiv [Preprint]. 2023 Nov 16:2023.11.14.567106. doi: 10.1101/2023.11.14.567106. bioRxiv. 2023. PMID: 38014215 Free PMC article. Preprint.
-
Effects of myokines on bone.Bonekey Rep. 2016 Jul 20;5:826. doi: 10.1038/bonekey.2016.48. eCollection 2016. Bonekey Rep. 2016. PMID: 27579164 Free PMC article. Review.
-
Targeting myostatin/activin A protects against skeletal muscle and bone loss during spaceflight.Proc Natl Acad Sci U S A. 2020 Sep 22;117(38):23942-23951. doi: 10.1073/pnas.2014716117. Epub 2020 Sep 8. Proc Natl Acad Sci U S A. 2020. PMID: 32900939 Free PMC article.
-
Activation of activin/Smad2 and 3 signaling pathway and the potential involvement of endothelial‑mesenchymal transition in the valvular damage due to rheumatic heart disease.Mol Med Rep. 2021 Jan;23(1):10. doi: 10.3892/mmr.2020.11648. Epub 2020 Nov 12. Mol Med Rep. 2021. PMID: 33179113 Free PMC article.
-
Differential bioactivity of four BMP-family members as function of biomaterial stiffness.Biomaterials. 2022 Feb;281:121363. doi: 10.1016/j.biomaterials.2022.121363. Epub 2022 Jan 4. Biomaterials. 2022. PMID: 35063741 Free PMC article.
References
-
- Olsen B. R., Reginato A. M., and Wang W. (2000) Bone development. Annu. Rev. Cell Dev. Biol. 16, 191–220 - PubMed
-
- Kablar B., Asakura A., Krastel K., Ying C., May L. L., Goldhamer D. J., and Rudnicki M. A. (1998) MyoD and Myf-5 define the specification of musculature of distinct embryonic origin. Biochem. Cell. Biol. 76, 1079–1091 - PubMed
-
- Hasty P., Bradley A., Morris J. H., Edmondson D. G., Venuti J. M., Olson E. N., and Klein W. H. (1993) Muscle deficiency and neonatal death in mice with a targeted mutation in the myogenin gene. Nature 364, 501–506 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

