Guanylyl cyclase sensitivity to nitric oxide is protected by a thiol oxidation-driven interaction with thioredoxin-1
- PMID: 28659344
- PMCID: PMC5582831
- DOI: 10.1074/jbc.M117.787390
Guanylyl cyclase sensitivity to nitric oxide is protected by a thiol oxidation-driven interaction with thioredoxin-1
Abstract
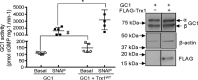
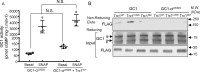
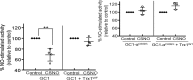
Nitric oxide (NO) modulates many physiological events through production of cGMP from its receptor, the NO-sensitive guanylyl cyclase (GC1). NO also appears to function in a cGMP-independent manner, via S-nitrosation (SNO), a redox-based modification of cysteine thiols. Previously, we have shown that S-nitrosated GC1 (SNO-GC1) is desensitized to NO stimulation following prolonged NO exposure or under oxidative/nitrosative stress. In animal models of nitrate tolerance and angiotensin II-induced hypertension, decreased vasodilation in response to NO correlates with GC1 thiol oxidation, but the physiological mechanism that resensitizes GC1 to NO and restores basal activity is unknown. Because GC1 interacts with the oxidoreductase protein-disulfide isomerase, we hypothesized that thioredoxin-1 (Trx1), a cytosolic oxidoreductase, could be involved in restoring GC1 basal activity and NO sensitivity because the Trx/thioredoxin reductase (TrxR) system maintains thiol redox homeostasis. Here, by manipulating activity and levels of the Trx1/TrxR system and by using a Trx1-Trap assay, we demonstrate that Trx1 modulates cGMP synthesis through an association between Trx1 and GC1 via a mixed disulfide. A proximity ligation assay confirmed the endogenous Trx1-GC1 complex in cells. Mutational analysis suggested that Cys609 in GC1 is involved in the Trx1-GC1 association and modulation of GC1 activity. Functionally, we established that Trx1 protects GC1 from S-nitrosocysteine-induced desensitization. A computational model of Trx1-GC1 interaction illustrates a possible mechanism for Trx1 to maintain basal GC1 activity and prevent/rescue GC1 desensitization to NO. The etiology of some oxidative vascular diseases may very well be explained by the dysfunction of the Trx1-GC1 association.
Keywords: S-nitrosylation; cyclic GMP (cGMP); guanylate cyclase (guanylyl cyclase); nitric oxide; oxidation-reduction (redox); thioredoxin.
© 2017 by The American Society for Biochemistry and Molecular Biology, Inc.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures




References
-
- Ignarro L. J. (2002) Nitric oxide as a unique signaling molecule in the vascular system: a historical overview. J. Physiol. Pharmacol. 53, 503–514 - PubMed
-
- Stamler J. S. (1994) Redox signaling: nitrosylation and related target interactions of nitric oxide. Cell 78, 931–936 - PubMed
-
- Hobbs A., Waldman S., Beuve A., Brouckaert P., Burnett J. C., Friebe A., Garthwaite J., Koesling D., Kuhn M., Potter L., and Schmidt H.. Guanylyl cyclases (GCs). Accessed on 02/08/17. IUPHAR/BPS Guide to Pharmacology. http://www.guidetopharmacology.org/GRAC/FamilyDisplayForward?familyId=258
-
- Takimoto E. (2012) Cyclic GMP-dependent signaling in cardiac myocytes. Circ. J. 76, 1819–1825 - PubMed
-
- Derbyshire E. R., and Marletta M. A. (2012) Structure and regulation of soluble guanylate cyclase. Annu. Rev. Biochem. 81, 533–559 - PubMed
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

