A Novel Theranostic Strategy for MMP-14-Expressing Glioblastomas Impacts Survival
- PMID: 28659432
- PMCID: PMC5587386
- DOI: 10.1158/1535-7163.MCT-17-0022
A Novel Theranostic Strategy for MMP-14-Expressing Glioblastomas Impacts Survival
Abstract
Glioblastoma (GBM) has a dismal prognosis. Evidence from preclinical tumor models and human trials indicates the role of GBM-initiating cells (GIC) in GBM drug resistance. Here, we propose a new treatment option with tumor enzyme-activatable, combined therapeutic and diagnostic (theranostic) nanoparticles, which caused specific toxicity against GBM tumor cells and GICs. The theranostic cross-linked iron oxide nanoparticles (CLIO) were conjugated to a highly potent vascular disrupting agent (ICT) and secured with a matrix-metalloproteinase (MMP-14) cleavable peptide. Treatment with CLIO-ICT disrupted tumor vasculature of MMP-14-expressing GBM, induced GIC apoptosis, and significantly impaired tumor growth. In addition, the iron core of CLIO-ICT enabled in vivo drug tracking with MR imaging. Treatment with CLIO-ICT plus temozolomide achieved tumor remission and significantly increased survival of human GBM-bearing mice by more than 2-fold compared with treatment with temozolomide alone. Thus, we present a novel therapeutic strategy with significant impact on survival and great potential for clinical translation. Mol Cancer Ther; 16(9); 1909-21. ©2017 AACR.
©2017 American Association for Cancer Research.
Figures
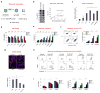

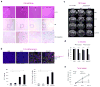

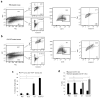

Comment on
-
Rethinking Brain Cancer Therapy: Tumor Enzyme Activatable Theranostic Nanoparticles.Mol Imaging. 2017 Jan-Dec;16:1536012117730950. doi: 10.1177/1536012117730950. Mol Imaging. 2017. PMID: 28929923 Free PMC article.
References
-
- Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352(10):987–96. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

