Glycosylation, an effective synthetic strategy to improve the bioavailability of therapeutic peptides
- PMID: 28660018
- PMCID: PMC5477030
- DOI: 10.1039/c5sc04392a
Glycosylation, an effective synthetic strategy to improve the bioavailability of therapeutic peptides
Abstract
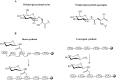
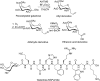
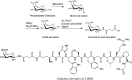
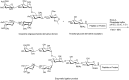
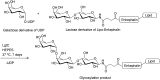
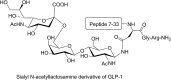
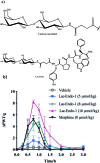
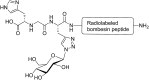
Glycosylation of peptides is a promising strategy for modulating the physicochemical properties of peptide drugs and for improving their absorption through biological membranes. This review highlights various methods for the synthesis of glycoconjugates and recent progress in the development of glycosylated peptide therapeutics. Furthermore, the impacts of glycosylation in overcoming the existing barriers that restrict oral and brain delivery of peptides are described herein.
Figures
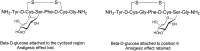
Similar articles
-
Chemoselective Solution- and Solid-Phase Synthesis of Disulfide-Linked Glycopeptides.J Org Chem. 2022 Nov 4;87(21):14026-14036. doi: 10.1021/acs.joc.2c01651. Epub 2022 Oct 20. J Org Chem. 2022. PMID: 36265181 Free PMC article.
-
Oral peptide delivery: Translational challenges due to physiological effects.J Control Release. 2018 Oct 10;287:167-176. doi: 10.1016/j.jconrel.2018.08.032. Epub 2018 Aug 23. J Control Release. 2018. PMID: 30145135 Review.
-
Oral delivery of peptide drugs: barriers and developments.BioDrugs. 2005;19(3):165-77. doi: 10.2165/00063030-200519030-00003. BioDrugs. 2005. PMID: 15984901 Review.
-
Optimization of physicochemical and pharmacological properties of peptide drugs by glycosylation.Methods Mol Biol. 2013;1081:107-36. doi: 10.1007/978-1-62703-652-8_8. Methods Mol Biol. 2013. PMID: 24014437 Review.
-
Glycosylated Cell-Penetrating Peptides (GCPPs).Chembiochem. 2019 Jun 3;20(11):1400-1409. doi: 10.1002/cbic.201800720. Epub 2019 Apr 12. Chembiochem. 2019. PMID: 30673159
Cited by
-
Genetically Encoded Quinone Methides Enabling Rapid, Site-Specific, and Photocontrolled Protein Modification with Amine Reagents.J Am Chem Soc. 2020 Oct 7;142(40):17057-17068. doi: 10.1021/jacs.0c06820. Epub 2020 Sep 25. J Am Chem Soc. 2020. PMID: 32915556 Free PMC article.
-
Peptide Release after Simulated Infant In Vitro Digestion of Dry Heated Cow's Milk Protein and Transport of Potentially Immunoreactive Peptides across the Caco-2 Cell Monolayer.Nutrients. 2020 Aug 18;12(8):2483. doi: 10.3390/nu12082483. Nutrients. 2020. PMID: 32824739 Free PMC article.
-
Synthesis of aryl-thioglycopeptides through chemoselective Pd-mediated conjugation.Chem Sci. 2018 Sep 20;9(46):8753-8759. doi: 10.1039/c8sc02370k. eCollection 2018 Dec 14. Chem Sci. 2018. PMID: 30627396 Free PMC article.
-
Positions of cysteine residues reveal local clusters and hidden relationships to Sequons and Transmembrane domains in Human proteins.Sci Rep. 2024 Oct 29;14(1):25886. doi: 10.1038/s41598-024-77056-8. Sci Rep. 2024. PMID: 39468182 Free PMC article.
-
Therapeutic peptides: chemical strategies fortify peptides for enhanced disease treatment efficacy.Amino Acids. 2025 May 8;57(1):25. doi: 10.1007/s00726-025-03454-5. Amino Acids. 2025. PMID: 40338379 Free PMC article. Review.
References
-
- Goodwin D., Simerska P., Toth I. Curr. Med. Chem. 2012;19:4451–4461. - PubMed
-
- Lalatsa A., in Fundamentals of Pharmaceutical Nanoscience, Springer, 2013, pp. 511–542.
LinkOut - more resources
Full Text Sources
Other Literature Sources

