Angiotensin II subtype 1a receptor signaling in resident hepatic macrophages induces liver metastasis formation
- PMID: 28660748
- PMCID: PMC5581524
- DOI: 10.1111/cas.13306
Angiotensin II subtype 1a receptor signaling in resident hepatic macrophages induces liver metastasis formation
Abstract
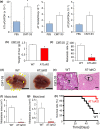
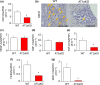
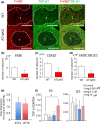
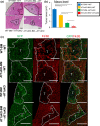
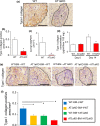
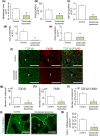
Liver metastases from colorectal cancer (CRC) are a clinically significant problem. The renin-angiotensin system is involved in tumor growth and metastases. This study was designed to evaluate the role of angiotensin II subtype receptor 1a (AT1a) in the formation of liver metastasis in CRC. A model of liver metastasis was developed by intrasplenic injection of mouse colon cancer (CMT-93) into AT1a knockout mice (AT1aKO) and wild-type (C57BL/6) mice (WT). Compared with WT mice, the liver weight and liver metastatic rate were significantly lower in AT1aKO. The mRNA levels of CD31, transforming growth factor- β1 (TGF-β1), and F4/80 were suppressed in AT1aKO compared with WT. Double immunofluorescence analysis showed that the number of accumulated F4/80+ cells expressing TGF-β1 in metastatic areas was higher in WT than in AT1aKO. The AT1aKO bone marrow (BM) (AT1aKO-BM)→WT showed suppressed formation of liver metastasis compared with WT-BM→WT. However, the formation of metastasis was further suppressed in WT-BM→AT1aKO compared with AT1aKO-BM→WT. In addition, accumulated F4/80+ cells in the liver metastasis were not BM-derived F4/80+ cells, but mainly resident hepatic F4/80+ cells, and these resident hepatic F4/80+ cells were positive for TGF-β1. Angiotensin II enhanced TGF-β1 expression in Kupffer cells. Treatment of WT with clodronate liposomes suppressed liver metastasis by diminishing TGF-β1+ F4/80+ cells accumulation. The formation of liver metastasis correlated with collagen deposition in the metastatic area, which was dependent on AT1a signaling. These results suggested that resident hepatic macrophages induced liver metastasis formation by induction of TGF-β1 through AT1a signaling.
Keywords: AT1a; Angiotensin; Kupffer cell; colorectal cancer; liver metastasis.
© 2017 The Authors. Cancer Science published by John Wiley & Sons Australia, Ltd on behalf of Japanese Cancer Association.
Figures


References
-
- Kono S. Secular trend of colon cancer incidence and mortality in relation to fat and meat intake in Japan. Eur J Cancer Prev 2004; 13: 127–32. - PubMed
-
- Fujimoto Y, Nakanishi Y, Sekine S et al CD10 expression in colorectal carcinoma correlates with liver metastasis. Dis Colon Rectum 2005; 48: 1839–89. - PubMed
-
- Itatani Y, Kawada K, Inamoto S et al The role of chemokines in promoting colorectal cancer invasion/metastasis. Int J Mol Sci 2016. (Apr 28);17: pii: E643 https://doi.org/10.3390/ijms17050643. - DOI - PMC - PubMed
-
- Bianchi G, Borgonovo G, Pistoia V et al Immunosuppressive cells and tumour microenvironment: focus on mesenchymal stem cells and myeloid derived suppressor cells. Histol Histopathol 2011; 26: 941–51. - PubMed
-
- Ochiumi Tl, Kitadai Y, Tanaka S et al Neuropilin‐1 is involved in regulation of apoptosis and migration of human colon cancer. Int J Oncol 2006; 29: 105–16. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

