Update: Influenza Activity in the United States During the 2016-17 Season and Composition of the 2017-18 Influenza Vaccine
- PMID: 28662019
- PMCID: PMC5687497
- DOI: 10.15585/mmwr.mm6625a3
Update: Influenza Activity in the United States During the 2016-17 Season and Composition of the 2017-18 Influenza Vaccine
Abstract
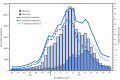
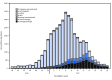
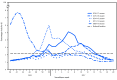
During the 2016-17 influenza season (October 2, 2016-May 20, 2017) in the United States, influenza activity* was moderate. Activity remained low through November, increased during December, and peaked in February nationally, although there were regional differences in the timing of influenza activity. Influenza A(H3N2) viruses predominated through mid-March and were predominant overall for the season, but influenza B viruses were most commonly reported from late March through May. This report summarizes influenza activity in the United States during October 2, 2016-May 20, 2017† and updates the previous summary (1).
Conflict of interest statement
Figures
References
-
- Food and Drug Administration. Summary minutes: meeting of the Vaccines and Related Biological Products Advisory Committee. Silver Spring, MD: US Department of Health and Human Services, Food and Drug Administration; 2017. https://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMateri...
-
- CDC. Disease burden of influenza. Atlanta, GA: US Department of Health and Human Services, CDC; 2017. https://www.cdc.gov/flu/about/disease/burden.htm
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

