An ABCC8 Nonsense Mutation Causing Neonatal Diabetes Through Altered Transcript Expression
- PMID: 28663158
- PMCID: PMC5596808
- DOI: 10.4274/jcrpe.4624
An ABCC8 Nonsense Mutation Causing Neonatal Diabetes Through Altered Transcript Expression
Abstract
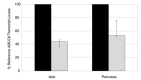
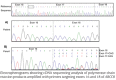
The pancreatic ATP-sensitive K+ (K-ATP) channel is a key regulator of insulin secretion. Gain-of-function mutations in the genes encoding the Kir6.2 (KCNJ11) and SUR1 (ABCC8) subunits of the channel cause neonatal diabetes, whilst loss-of-function mutations in these genes result in congenital hyperinsulinism. We report two patients with neonatal diabetes in whom we unexpectedly identified recessively inherited loss-of-function mutations. The aim of this study was to investigate how a homozygous nonsense mutation in ABCC8 could result in neonatal diabetes. The ABCC8 p.Glu747* was identified in two unrelated Vietnamese patients. This mutation is located within the in-frame exon 17 and RNA studies confirmed (a) the absence of full length SUR1 mRNA and (b) the presence of the alternatively spliced transcript lacking exon 17. Successful transfer of both patients to sulphonylurea treatment suggests that the altered transcript expression enhances the sensitivity of the K-ATP channel to Mg-ADP/ATP. This is the first report of an ABCC8 nonsense mutation causing a gain-of-channel function and these findings extend the spectrum of K-ATP channel mutations observed in patients with neonatal diabetes.
Keywords: Neonatal diabetes; nonsense mutation splicing..
Figures
Similar articles
-
Update of variants identified in the pancreatic β-cell KATP channel genes KCNJ11 and ABCC8 in individuals with congenital hyperinsulinism and diabetes.Hum Mutat. 2020 May;41(5):884-905. doi: 10.1002/humu.23995. Epub 2020 Feb 17. Hum Mutat. 2020. PMID: 32027066 Free PMC article. Review.
-
Functional characterization of activating mutations in the sulfonylurea receptor 1 (ABCC8) causing neonatal diabetes mellitus in Asian Indian children.Pediatr Diabetes. 2019 Jun;20(4):397-407. doi: 10.1111/pedi.12843. Epub 2019 Apr 2. Pediatr Diabetes. 2019. PMID: 30861254 Free PMC article.
-
ABCC8 and KCNJ11 molecular spectrum of 109 patients with diazoxide-unresponsive congenital hyperinsulinism.J Med Genet. 2010 Nov;47(11):752-9. doi: 10.1136/jmg.2009.075416. Epub 2010 Aug 3. J Med Genet. 2010. PMID: 20685672
-
A novel mutation in ABCC8 gene in a newborn with congenital hyperinsulinism -a case report.Fetal Pediatr Pathol. 2013 Dec;32(6):412-7. doi: 10.3109/15513815.2013.789947. Epub 2013 Apr 22. Fetal Pediatr Pathol. 2013. PMID: 23607867
-
New insights into KATP channel gene mutations and neonatal diabetes mellitus.Nat Rev Endocrinol. 2020 Jul;16(7):378-393. doi: 10.1038/s41574-020-0351-y. Epub 2020 May 6. Nat Rev Endocrinol. 2020. PMID: 32376986 Review.
Cited by
-
Clinical and Genetic Characteristics of ABCC8 Nonneonatal Diabetes Mellitus: A Systematic Review.J Diabetes Res. 2021 Sep 30;2021:9479268. doi: 10.1155/2021/9479268. eCollection 2021. J Diabetes Res. 2021. PMID: 34631896 Free PMC article.
-
Birth weight and diazoxide unresponsiveness strongly predict the likelihood of congenital hyperinsulinism due to a mutation in ABCC8 or KCNJ11.Eur J Endocrinol. 2021 Oct 30;185(6):813-818. doi: 10.1530/EJE-21-0476. Eur J Endocrinol. 2021. PMID: 34633981 Free PMC article.
-
The Elusive Nature of ABCC8-related Maturity-Onset Diabetes of the Young (ABCC8-MODY). A Review of the Literature and Case Discussion.Curr Diab Rep. 2024 Sep;24(9):197-206. doi: 10.1007/s11892-024-01547-1. Epub 2024 Jul 9. Curr Diab Rep. 2024. PMID: 38980630 Free PMC article. Review.
-
Molecular Genetics, Clinical Characteristics, and Treatment Outcomes of KATP-Channel Neonatal Diabetes Mellitus in Vietnam National Children's Hospital.Front Endocrinol (Lausanne). 2021 Sep 9;12:727083. doi: 10.3389/fendo.2021.727083. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 34566892 Free PMC article.
-
The Genetic Spectrum of Maturity-Onset Diabetes of the Young (MODY) in Qatar, a Population-Based Study.Int J Mol Sci. 2022 Dec 21;24(1):130. doi: 10.3390/ijms24010130. Int J Mol Sci. 2022. PMID: 36613572 Free PMC article.
References
-
- Flanagan SE, Clauin S, Bellanne-Chantelot C, Lonlay P, de, Harries LW, Gloyn AL, Ellard S. Update of mutations in the genes encoding the pancreatic beta-cell K(ATP) channel subunits Kir6.2 (KCNJ11) and sulfonylurea receptor 1 (ABCC8) in diabetes mellitus and hyperinsulinism. Human mutation. 2009;30:170–180. - PubMed
-
- Taschenberger G, Mougey A, Shen S, Lester LB, LaFranchi S, Shyng SL. Identification of a familial hyperinsulinism-causing mutation in the sulfonylurea receptor 1 that prevents normal trafficking and function of KATP channels. J Biol Chem. 2002;277:17139–17146. - PubMed
-
- Huopio H, Shyng SL, Otonkoski T, Nichols CG. K(ATP) channels and insulin secretion disorders. Am J Physiol Endocrinol Metab. 2002;283:207–216. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
