PATHway I: design and rationale for the investigation of the feasibility, clinical effectiveness and cost-effectiveness of a technology-enabled cardiac rehabilitation platform
- PMID: 28667228
- PMCID: PMC5726129
- DOI: 10.1136/bmjopen-2017-016781
PATHway I: design and rationale for the investigation of the feasibility, clinical effectiveness and cost-effectiveness of a technology-enabled cardiac rehabilitation platform
Abstract
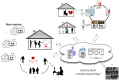
Introduction: Exercise-based cardiac rehabilitation (CR) independently alters the clinical course of cardiovascular diseases resulting in a significant reduction in all-cause and cardiac mortality. However, only 15%-30% of all eligible patients participate in a phase 2 ambulatory programme. The uptake rate of community-based programmes following phase 2 CR and adherence to long-term exercise is extremely poor. Newer care models, involving telerehabilitation programmes that are delivered remotely, show considerable promise for increasing adherence. In this view, the PATHway (Physical Activity Towards Health) platform was developed and now needs to be evaluated in terms of its feasibility and clinical efficacy.
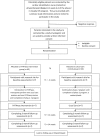
Methods and analysis: In a multicentre randomised controlled pilot trial, 120 participants (m/f, age 40-80 years) completing a phase 2 ambulatory CR programme will be randomised on a 1:1 basis to PATHway or usual care. PATHway involves a comprehensive, internet-enabled, sensor-based home CR platform and provides individualised heart rate monitored exercise programmes (exerclasses and exergames) as the basis on which to provide a personalised lifestyle intervention programme. The control group will receive usual care. Study outcomes will be assessed at baseline, 3 months and 6 months after completion of phase 2 of the CR programme. The primary outcome is the change in active energy expenditure. Secondary outcomes include cardiopulmonary endurance capacity, muscle strength, body composition, cardiovascular risk factors, peripheral endothelial vascular function, patient satisfaction, health-related quality of life (HRQoL), well-being, mediators of behaviour change and safety. HRQoL and healthcare costs will be taken into account in cost-effectiveness evaluation.
Ethics and dissemination: The study will be conducted in accordance with the Declaration of Helsinki. This protocol has been approved by the director and clinical director of the PATHway study and by the ethical committee of each participating site. Results will be disseminated via peer-reviewed scientific journals and presentations at congresses and events.
Trial registration number: NCT02717806. This trial is currently in the pre-results stage.
Keywords: cardiac rehabilitation; design; exercise; randomized controlled trial; telemonitoring; telerehabilitation.
© Article author(s) (or their employer(s) unless otherwise stated in the text of the article) 2017. All rights reserved. No commercial use is permitted unless otherwise expressly granted.
Conflict of interest statement
Competing interests: None declared.
Figures
References
-
- Cardiology EHNaESo. European Cardiovascular Disease Statistics. 2012. http://www.ehnheart.org/cvd-statistics/cvd-statistics-2012.html
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
