Fatty acid transport protein 1 regulates retinoid metabolism and photoreceptor development in mouse retina
- PMID: 28672005
- PMCID: PMC5495297
- DOI: 10.1371/journal.pone.0180148
Fatty acid transport protein 1 regulates retinoid metabolism and photoreceptor development in mouse retina
Abstract
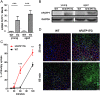
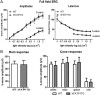
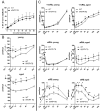
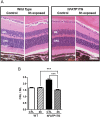
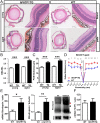
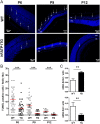
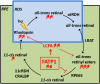
In retinal pigment epithelium (RPE), RPE65 catalyzes the isomerization of all-trans-retinyl fatty acid esters to 11-cis-retinol in the visual cycle and controls the rhodopsin regeneration rate. However, the mechanisms by which these processes are regulated are still unclear. Fatty Acid Transport Protein 1 (FATP1) is involved in fatty acid uptake and lipid metabolism in a variety of cell types. FATP1 co-localizes with RPE65 in RPE and inhibits its isomerase activity in vitro. Here, we further investigated the role of FATP1 in the visual cycle using transgenic mice that overexpress human FATP1 specifically in the RPE (hFATP1TG mice). The mice displayed no delay in the kinetics of regeneration of the visual chromophore 11-cis-retinal after photobleaching and had no defects in light sensitivity. However, the total retinoid content was higher in the hFATP1TG mice than in wild type mice, and the transgenic mice also displayed an age-related accumulation (up to 40%) of all-trans-retinal and retinyl esters that was not observed in control mice. Consistent with these results, hFATP1TG mice were more susceptible to light-induced photoreceptor degeneration. hFATP1 overexpression also induced an ~3.5-fold increase in retinosome autofluorescence, as measured by two-photon microscopy. Interestingly, hFATP1TG retina contained ~25% more photoreceptor cells and ~35% longer outer segments than wild type mice, revealing a non-cell-autonomous effect of hFATP1 expressed in the RPE. These data are the first to show that FATP1-mediated fatty acid uptake in the RPE controls both retinoid metabolism in the outer retina and photoreceptor development.
Conflict of interest statement
Figures

References
-
- Bretillon L, Thuret G, Gregoire S, Acar N, Joffre C, et al. (2008) Lipid and fatty acid profile of the retina, retinal pigment epithelium/choroid, and the lacrimal gland, and associations with adipose tissue fatty acids in human subjects. Exp Eye Res 87: 521–528. doi: 10.1016/j.exer.2008.08.010 - DOI - PubMed
-
- Wright CB, Redmond TM, Nickerson JM (2015) A History of the Classical Visual Cycle. Prog Mol Biol Transl Sci 134: 433–448. doi: 10.1016/bs.pmbts.2015.06.009 - DOI - PubMed
-
- Sears AE, Palczewski K (2016) Lecithin:Retinol Acyltransferase: A Key Enzyme Involved in the Retinoid (visual) Cycle. Biochemistry 55: 3082–3091. doi: 10.1021/acs.biochem.6b00319 - DOI - PMC - PubMed
-
- Jin M, Li S, Moghrabi WN, Sun H, Travis GH (2005) Rpe65 is the retinoid isomerase in bovine retinal pigment epithelium. Cell 122: 449–459. doi: 10.1016/j.cell.2005.06.042 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

