ω-3 Polyunsaturated fatty acids and their cytochrome P450-derived metabolites suppress colorectal tumor development in mice
- PMID: 28672272
- PMCID: PMC6278818
- DOI: 10.1016/j.jnutbio.2017.06.006
ω-3 Polyunsaturated fatty acids and their cytochrome P450-derived metabolites suppress colorectal tumor development in mice
Abstract
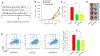
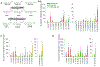
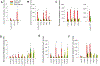
Many studies have shown that dietary intake of ω-3 polyunsaturated fatty acids (PUFAs) reduces the risks of colorectal cancer; however, the underlying mechanisms are not well understood. Here we used a LC-MS/MS-based lipidomics to explore the role of eicosanoid signaling in the anti-colorectal cancer effects of ω-3 PUFAs. Our results showed that dietary feeding of ω-3 PUFAs-rich diets suppressed growth of MC38 colorectal tumor, and modulated profiles of fatty acids and eicosanoid metabolites in C57BL/6 mice. Notably, we found that dietary feeding of ω-3 PUFAs significantly increased levels of epoxydocosapentaenoic acids (EDPs, metabolites of ω-3 PUFA produced by cytochrome P450 enzymes) in plasma and tumor tissue of the treated mice. We further showed that systematic treatment with EDPs (dose=0.5 mg/kg per day) suppressed MC38 tumor growth in mice, with reduced expressions of pro-oncogenic genes such as C-myc, Axin2, and C-jun in tumor tissues. Together, these results support that formation of EDPs might contribute to the anti-colorectal cancer effects of ω-3 PUFAs.
Keywords: Colorectal cancer; Docosahexaenoic acid (DHA); Epoxydocosapentaenoic acids (EDPs); Lipidomics; ω-3 Polyunsaturated fatty acids (PUFAs).
Copyright © 2017 Elsevier Inc. All rights reserved.
Figures

References
-
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66:7–30. - PubMed
-
- Sasazuki S, Inoue M, Iwasaki M, Sawada N, Shimazu T, Yamaji T, et al. Intake of n-3 and n-6 polyunsaturated fatty acids and development of colorectal cancer by subsite: Japan Public Health Center-based prospective study. Int J Cancer. 2011;129:1718–29. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

