Rutin suppresses high glucose-induced ACTA2 and p38 protein expression in diabetic nephropathy
- PMID: 28672912
- PMCID: PMC5488511
- DOI: 10.3892/etm.2017.4509
Rutin suppresses high glucose-induced ACTA2 and p38 protein expression in diabetic nephropathy
Abstract
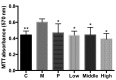
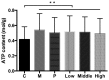
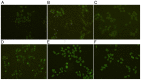
The present study investigated the effect of rutin on high glucose-induced actin, α2, smooth muscle, aorta (ACTA2) and p38 protein expression in diabetic nephropathy (DN). Human mesangial cells were divided into a control group, high glucose-induced mesangial cell group, high glucose + captopril group, and high glucose + rutin group (low, middle and high doses of rutin). Cell viability, adenosine 5'-triphosphate (ATP) content, cell cycle, and ACTA2 and p38 protein expression were examined using MTT assay, ATP assay kit, flow cytometry and immunofluorescence staining in cultured human mesangial cells, respectively. Cell viability, ATP content, and ACTA2 and p38 expression increased significantly in high glucose-induced mesangial cells (P<0.05). However, at concentrations of 0.2, 0.4 and 0.8 µmol/l rutin was able to inhibit high glucose-induced human mesangial cell viability, ATP content, and ACTA2 and p38 expression and improve the cell cycle progression of mesangial cells. In conclusion, ACTA2 and p38 proteins may have important roles in DN. Rutin may inhibit the expression of ACTA2 and p38 and may be utilized in the prevention and treatment of DN.
Keywords: actin; aorta; diabetic nephropathy; human mesangial cells; p38; rutin; smooth muscle; α2.
Figures



Similar articles
-
Protective effects of rutin on rat glomerular mesangial cells cultured in high glucose conditions.Phytother Res. 2011 Nov;25(11):1640-7. doi: 10.1002/ptr.3461. Epub 2011 Mar 21. Phytother Res. 2011. Retraction in: Phytother Res. 2013 Oct;27(10):1580. PMID: 21425374 Retracted.
-
The anti-inflammation effect of Moutan Cortex on advanced glycation end products-induced rat mesangial cells dysfunction and High-glucose-fat diet and streptozotocin-induced diabetic nephropathy rats.J Ethnopharmacol. 2014;151(1):591-600. doi: 10.1016/j.jep.2013.11.015. Epub 2013 Nov 21. J Ethnopharmacol. 2014. PMID: 24269777
-
Preventive effects of rutin on the development of experimental diabetic nephropathy in rats.Life Sci. 2012 Nov 2;91(19-20):959-67. doi: 10.1016/j.lfs.2012.09.003. Epub 2012 Sep 20. Life Sci. 2012. PMID: 23000098
-
[Dracorhodin perchlorate inhibit high glucose induce serum and glucocorticoid induced protein kinase 1 and fibronectin expression in human mesangial cells].Zhongguo Zhong Yao Za Zhi. 2010 Aug;35(15):1996-2000. doi: 10.4268/cjcmm20101518. Zhongguo Zhong Yao Za Zhi. 2010. PMID: 20931854 Chinese.
-
[Phenotypic and functional alterations of mesangial cells in patients with diabetic nephropathy].Zhonghua Yi Xue Za Zhi. 2001 Nov 25;81(22):1369-73. Zhonghua Yi Xue Za Zhi. 2001. PMID: 11930630 Chinese.
Cited by
-
System Pharmacology-Based Dissection of the Synergistic Mechanism of Huangqi and Huanglian for Diabetes Mellitus.Front Pharmacol. 2017 Oct 5;8:694. doi: 10.3389/fphar.2017.00694. eCollection 2017. Front Pharmacol. 2017. PMID: 29051733 Free PMC article.
-
Oxidative Stress and NRF2/KEAP1/ARE Pathway in Diabetic Kidney Disease (DKD): New Perspectives.Biomolecules. 2022 Sep 2;12(9):1227. doi: 10.3390/biom12091227. Biomolecules. 2022. PMID: 36139066 Free PMC article. Review.
-
Flavonoids on diabetic nephropathy: advances and therapeutic opportunities.Chin Med. 2021 Aug 7;16(1):74. doi: 10.1186/s13020-021-00485-4. Chin Med. 2021. PMID: 34364389 Free PMC article. Review.
-
Decoding the differentiation of mesenchymal stem cells into mesangial cells at the transcriptomic level.BMC Genomics. 2020 Jul 7;21(1):467. doi: 10.1186/s12864-020-06868-5. BMC Genomics. 2020. PMID: 32635896 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
