Cytokine-induced killer cell therapy for modulating regulatory T cells in patients with non-small cell lung cancer
- PMID: 28673007
- PMCID: PMC5488714
- DOI: 10.3892/etm.2017.4562
Cytokine-induced killer cell therapy for modulating regulatory T cells in patients with non-small cell lung cancer
Abstract
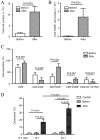
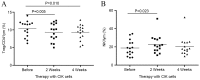
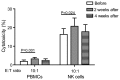
Previous studies have reported that regulatory T cells (Tregs), which are physiologically engaged in the maintenance of immunological self-tolerance, have a critical role in the regulation of the antitumor immune response. Targeting Tregs has the potential to augment cancer vaccine approaches. The current study therefore aimed to evaluate the role of cytokine-induced killer (CIK) cell infusion in modulating Tregs in patients with non-small cell lung cancer (NSCLC). A total of 15 patients with advanced NSCLC were treated by an infusion of CIK cells derived from autologous peripheral blood mononuclear cells (PBMCs). By using flow cytometry and liquid chip analysis, subsets of T cells and natural killer (NK) cells in peripheral blood, and plasma cytokine profiles in the treated patients, were analyzed at 2 and 4 weeks after CIK cell infusion. Cytotoxicity of PBMCs (n=15) and NK cells (n=6) isolated from NSCLC patients was evaluated before and after CIK cell therapy. Progression-free survival (PFS) and overall survival (OS) were also assessed. Analysis of the immune cell populations before and after treatment showed a significant increase in NK cells (P<0.05) concomitant with a significant decrease in Tregs (P<0.01) at 2 weeks post-infusion of CIK cells compared with the baseline. NK group 2D receptor (NKG2D) expression on NK cells was also significantly increased at 2 weeks post-infusion compared with the baseline (P<0.05). There was a positive correlation between NKG2D expression and the infusion number of CIK cells (P<0.05). When evaluated at 2 weeks after CIK cell therapy, the cytotoxicity of PBMCs and isolated NK cells was significantly increased compared with the baseline (P<0.01 and P<0.05). Correspondingly, plasma cytokine profiles showed significant enhancement of the following antitumor cytokines: Interferon (IFN)-γ (P<0.05), IFN-γ-inducible protein 10 (P<0.01), tumor necrosis factor-α (P<0.001), granulocyte-macrophage colony-stimulating factor (P<0.01), monocyte chemotactic protein-3 (P<0.01) and interleukin-21 (P<0.05) at 2 weeks post-infusion, compared with the baseline. At the same time, the expression of transforming growth factor-β1, which is primarily produced by Tregs, was significantly decreased compared with the baseline (P<0.05). Median PFS and OS in the CIK cell treatment group were significantly increased compared with the control group (PFS, 9.98 vs. 5.44 months, P=0.038; OS, 24.17 vs. 20.19 months, P=0.048). No severe side-effects were observed during the treatment period. In conclusion, CIK cell therapy was able to suppress Tregs and enhance the antitumor immunity of NK cells in advanced NSCLC patients. Therefore, CIK cell treatment may improve PFS and OS in patients with advanced NSCLC. CIK cell infusion may have therapeutic value for patients with advanced NSCLC, as a treatment that can be combined with chemotherapy and radiotherapy.
Keywords: cytokine-induced killer cells; non-small cell lung cancer; regulatory T cells.
Figures



Similar articles
-
Combination of DC/CIK adoptive T cell immunotherapy with chemotherapy in advanced non-small-cell lung cancer (NSCLC) patients: a prospective patients' preference-based study (PPPS).Clin Transl Oncol. 2019 Jun;21(6):721-728. doi: 10.1007/s12094-018-1968-3. Epub 2018 Oct 29. Clin Transl Oncol. 2019. PMID: 30374838
-
Clinical efficacy and safety of CIK plus radiotherapy for lung cancer: A meta-analysis of 16 randomized controlled trials.Int Immunopharmacol. 2018 Aug;61:363-375. doi: 10.1016/j.intimp.2018.06.012. Epub 2018 Jun 23. Int Immunopharmacol. 2018. PMID: 29945024
-
Influence of the number and interval of treatment cycles on cytokine-induced killer cells and their adjuvant therapeutic effects in advanced non-small-cell lung cancer (NSCLC).Int Immunopharmacol. 2017 Sep;50:263-269. doi: 10.1016/j.intimp.2017.07.006. Epub 2017 Jul 12. Int Immunopharmacol. 2017. PMID: 28711032
-
Effectiveness and safety of chemotherapy with cytokine-induced killer cells in non-small cell lung cancer: A systematic review and meta-analysis of 32 randomized controlled trials.Cytotherapy. 2019 Feb;21(2):125-147. doi: 10.1016/j.jcyt.2018.10.011. Epub 2018 Dec 14. Cytotherapy. 2019. PMID: 30554868
-
Cytokine-induced killer cells: NK-like T cells with cytotolytic specificity against leukemia.Leuk Lymphoma. 2003 Sep;44(9):1457-62. doi: 10.3109/10428190309178764. Leuk Lymphoma. 2003. PMID: 14565644 Review.
Cited by
-
The expression level of CSDAP1 in lung cancer and its clinical significance.Oncol Lett. 2018 Oct;16(4):4361-4366. doi: 10.3892/ol.2018.9195. Epub 2018 Jul 23. Oncol Lett. 2018. PMID: 30214570 Free PMC article.
-
Complete Response to PD-1 Inhibitor in Primary Hepatocellular Carcinoma Patients Post-Progression on Bi-Specific Antibody Conjugated CIK Cell Treatment: A Report of Two Cases.Onco Targets Ther. 2021 Dec 18;14:5447-5453. doi: 10.2147/OTT.S333604. eCollection 2021. Onco Targets Ther. 2021. PMID: 34984004 Free PMC article.
-
Retrospective analysis of the efficacy of adjuvant cytokine-induced killer cell immunotherapy combined with chemotherapy in colorectal cancer patients after surgery.Clin Transl Immunology. 2022 Jan 18;11(1):e1368. doi: 10.1002/cti2.1368. eCollection 2022. Clin Transl Immunology. 2022. PMID: 35079378 Free PMC article.
-
Ultrasound-mediated microbubble destruction: a new method in cancer immunotherapy.Onco Targets Ther. 2018 Sep 12;11:5763-5775. doi: 10.2147/OTT.S171019. eCollection 2018. Onco Targets Ther. 2018. PMID: 30254469 Free PMC article. Review.
-
Ex vivo-expanded highly purified natural killer cells in combination with temozolomide induce antitumor effects in human glioblastoma cells in vitro.PLoS One. 2019 Mar 6;14(3):e0212455. doi: 10.1371/journal.pone.0212455. eCollection 2019. PLoS One. 2019. PMID: 30840664 Free PMC article.
References
-
- Farashi-Bonab S, Khansari N. Regulatory T cells in cancer patients and their roles in cancer development/progression. MOJ Immunol. 2014;1 00024.
LinkOut - more resources
Full Text Sources
Other Literature Sources
