Host-mediated impairment of parasite maturation during blood-stage Plasmodium infection
- PMID: 28673996
- PMCID: PMC5530648
- DOI: 10.1073/pnas.1618939114
Host-mediated impairment of parasite maturation during blood-stage Plasmodium infection
Abstract
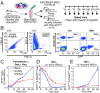
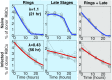
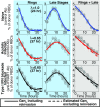
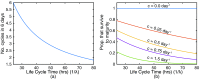
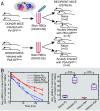
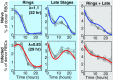
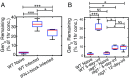
Severe malaria and associated high parasite burdens occur more frequently in humans lacking robust adaptive immunity to Plasmodium falciparum Nevertheless, the host may partly control blood-stage parasite numbers while adaptive immunity is gradually established. Parasite control has typically been attributed to enhanced removal of parasites by the host, although in vivo quantification of this phenomenon remains challenging. We used a unique in vivo approach to determine the fate of a single cohort of semisynchronous, Plasmodium berghei ANKA- or Plasmodium yoelii 17XNL-parasitized red blood cells (pRBCs) after transfusion into naive or acutely infected mice. As previously shown, acutely infected mice, with ongoing splenic and systemic inflammatory responses, controlled parasite population growth more effectively than naive controls. Surprisingly, however, this was not associated with accelerated removal of pRBCs from circulation. Instead, transfused pRBCs remained in circulation longer in acutely infected mice. Flow cytometric assessment and mathematical modeling of intraerythrocytic parasite development revealed an unexpected and substantial slowing of parasite maturation in acutely infected mice, extending the life cycle from 24 h to 40 h. Importantly, impaired parasite maturation was the major contributor to control of parasite growth in acutely infected mice. Moreover, by performing the same experiments in rag1-/- mice, which lack T and B cells and mount weak inflammatory responses, we revealed that impaired parasite maturation is largely dependent upon the host response to infection. Thus, impairment of parasite maturation represents a host-mediated, immune system-dependent mechanism for limiting parasite population growth during the early stages of an acute blood-stage Plasmodium infection.
Keywords: Plasmodium berghei ANKA; clearance; malaria; mathematical modelling; parasite maturation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Characterising the effect of antimalarial drugs on the maturation and clearance of murine blood-stage Plasmodium parasites in vivo.Int J Parasitol. 2017 Dec;47(14):913-922. doi: 10.1016/j.ijpara.2017.05.009. Epub 2017 Aug 31. Int J Parasitol. 2017. PMID: 28864033
-
Mosquito immune responses and compatibility between Plasmodium parasites and anopheline mosquitoes.BMC Microbiol. 2009 Jul 30;9:154. doi: 10.1186/1471-2180-9-154. BMC Microbiol. 2009. PMID: 19643026 Free PMC article.
-
Febrile temperature but not proinflammatory cytokines promotes phosphatidylserine expression on Plasmodium falciparum malaria-infected red blood cells during parasite maturation.Cytometry A. 2010 Jun;77(6):515-23. doi: 10.1002/cyto.a.20879. Cytometry A. 2010. PMID: 20191617
-
Defence mechanisms and immune evasion in the interplay between the humane immune system and Plasmodium falciparum.Dan Med Bull. 1992 Feb;39(1):49-63. Dan Med Bull. 1992. PMID: 1563295 Review.
-
Selected problems of malaria blood stage immunity.Tokai J Exp Clin Med. 1998 Apr;23(2):55-62. Tokai J Exp Clin Med. 1998. PMID: 10021776 Review.
Cited by
-
Stopping Plasmodium Parasite Invasion by Boosting Humoral Immunity.Hemasphere. 2019 Jun 4;3(3):e206. doi: 10.1097/HS9.0000000000000206. eCollection 2019 Jun. Hemasphere. 2019. PMID: 31723834 Free PMC article. No abstract available.
-
In Silico Investigation of the Decline in Clinical Efficacy of Artemisinin Combination Therapies Due to Increasing Artemisinin and Partner Drug Resistance.Antimicrob Agents Chemother. 2018 Nov 26;62(12):e01292-18. doi: 10.1128/AAC.01292-18. Print 2018 Dec. Antimicrob Agents Chemother. 2018. PMID: 30249691 Free PMC article.
-
The Plasmodium falciparum transcriptome in severe malaria reveals altered expression of genes involved in important processes including surface antigen-encoding var genes.PLoS Biol. 2018 Mar 12;16(3):e2004328. doi: 10.1371/journal.pbio.2004328. eCollection 2018 Mar. PLoS Biol. 2018. PMID: 29529020 Free PMC article.
-
Methodology to streamline flow cytometric-based detection of early stage Plasmodium parasitemia in mice.J Microbiol Methods. 2022 Apr;195:106439. doi: 10.1016/j.mimet.2022.106439. Epub 2022 Mar 4. J Microbiol Methods. 2022. PMID: 35248600 Free PMC article.
-
Parasite-Host Dynamics throughout Antimalarial Drug Development Stages Complicate the Translation of Parasite Clearance.Antimicrob Agents Chemother. 2021 Mar 18;65(4):e01539-20. doi: 10.1128/AAC.01539-20. Print 2021 Mar 18. Antimicrob Agents Chemother. 2021. PMID: 33526486 Free PMC article.
References
-
- Molineaux L, Träuble M, Collins WE, Jeffery GM, Dietz K. Malaria therapy reinoculation data suggest individual variation of an innate immune response and independent acquisition of antiparasitic and antitoxic immunities. Trans R Soc Trop Med Hyg. 2002;96:205–209. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

