Intratumoral STING Activation with T-cell Checkpoint Modulation Generates Systemic Antitumor Immunity
- PMID: 28674082
- PMCID: PMC5547907
- DOI: 10.1158/2326-6066.CIR-17-0049
Intratumoral STING Activation with T-cell Checkpoint Modulation Generates Systemic Antitumor Immunity
Abstract
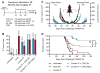
Coordinated manipulation of independent immune regulatory pathways in the tumor microenvironment-including blockade of T-cell checkpoint receptors and reversal of suppressive myeloid programs-can render aggressive cancers susceptible to immune rejection. Elevated toxicity associated with combination immunotherapy, however, prevents translation of the most efficacious regimens. We evaluated T-cell checkpoint-modulating antibodies targeting CTLA-4, PD-1, and 4-1BB together with myeloid agonists targeting either STING or Flt3 in the TRAMP-C2 model of prostate cancer to determine whether low-dose intratumoral delivery of these agents could elicit systemic control of multifocal disease. Intratumoral administration of the STING agonist cyclic di-GMP (CDG) or Flt3 Ligand (Flt3L) augmented the therapeutic effect of systemic triple checkpoint modulation and promoted the cure of 75% of mice with bilateral TRAMP-C2; however, when all agents were administered locally, only CDG mobilized abscopal immunity. Combination efficacy correlated with globally enhanced ratios of CD8+ T cells to regulatory T cells (Treg), macrophages, and myeloid-derived suppressor cells, and downregulation of the M2 marker CD206 on tumor-associated macrophages. Flt3L improved CD8+ T-cell and dendritic cell infiltration of tumors, but was diminished in efficacy by concomitant Treg expansion. Although intratumoral CDG/checkpoint therapy invokes substantial ulceration at the injection site, reduced CDG dosing can preserve tissue integrity without sacrificing therapeutic benefit. For high-order combinations of T-cell checkpoint antibodies and local myeloid agonists, systemic antibody administration provides the greatest efficacy; however, local administration of CDG and antibody provides substantial systemic benefit while minimizing the potential for immune-related adverse events. Cancer Immunol Res; 5(8); 676-84. ©2017 AACR.
©2017 American Association for Cancer Research.
Conflict of interest statement
Figures





References
-
- Kwon ED, Drake CG, Scher HI, Fizazi K, Bossi A, van den Eertwegh AJM, et al. Ipilimumab versus placebo after radiotherapy in patients with metastatic castration-resistant prostate cancer that had progressed after docetaxel chemotherapy (CA184-043): a multicentre, randomised, double-blind, phase 3 trial. The Lancet Oncology. 15(7):700–12. doi: 10.1016/S1470-2045(14)70189-5. - DOI - PMC - PubMed
-
- Foster BA, Gingrich JR, Kwon ED, Madias C, Greenberg NM. Characterization of prostatic epithelial cell lines derived from transgenic adenocarcinoma of the mouse prostate (TRAMP) model. Cancer research. 1997;57(16):3325–30. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

