Investigating the safety and activity of the use of BTT1023 (Timolumab), in the treatment of patients with primary sclerosing cholangitis (BUTEO): A single-arm, two-stage, open-label, multi-centre, phase II clinical trial protocol
- PMID: 28674140
- PMCID: PMC5734279
- DOI: 10.1136/bmjopen-2016-015081
Investigating the safety and activity of the use of BTT1023 (Timolumab), in the treatment of patients with primary sclerosing cholangitis (BUTEO): A single-arm, two-stage, open-label, multi-centre, phase II clinical trial protocol
Abstract
Introduction: Primary sclerosing cholangitis (PSC) is a progressive inflammatory liver disease characterised by relentless liver fibrosis and a high unmet need for new therapies. Preventing fibrosis represents an important area of interest in the development of vital new drugs. Vascular adhesion protein-1 (VAP-1) drives inflammation in liver disease, and provision of an antibody against VAP-1 blunts fibrosis in murine models of liver injury.
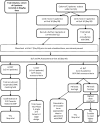
Methods and analysis: BUTEO is a single-arm, two-stage, open-label, multi-centre, phase II clinical trial. Up to 59 patients will receive treatment with anti-VAP monoclonal antibody, BTT1023, over a 78-day treatment period. Adults with PSC and a serum alkaline phosphatase (ALP) of at least 1.5 times the upper limit of normal will be included. Our primary outcome measure is a reduction in ALP by >25% from baseline to Day 99. Secondary outcome measures include safety and tolerability, changes pre therapy/post therapy in circulating serum VAP-1 as well as imaging findings. The first patient participant was recruited on 08 September 2015.
Ethics and dissemination: This protocol has been approved by the Research Ethics Committee (REC, reference 14/EM/1272). The first REC approval date was 06 January 2015 with three subsequent approved amendments. This article refers to protocol V3.0, dated 16 March 2016. Results will be disseminated via peer-reviewed publication and presentation at international conferences.
Trial registration: The trial is registered with the European Medicines agency (EudraCT: 2014-002393-37), the National Institute for Health Research (Portfolio ID: 18051) and ISRCTN: 11233255. The clinicaltrials.gov identifier is NCT02239211. Pre-results.
Keywords: Clinical Trials; Hepatobiliary Disease; Hepatology; Immunology.
© Article author(s) (or their employer(s) unless otherwise stated in the text of the article) 2017. All rights reserved. No commercial use is permitted unless otherwise expressly granted.
Conflict of interest statement
Competing interests: CW and DHA report grants from NIHR MRC, during the conduct of thestudy; grants and non-financial support from Pharmaxis Inc, Australia,outside the submitted work; and have a patent "The use of VAP-1 inhibitors for treating fibroticconditions (WO 2011029996 A1)" issued. Other authors have no competing interests declared.
Figures
References
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials