Investigating β-adrenergic-induced cardiac hypertrophy through computational approach: classical and non-classical pathways
- PMID: 28674776
- PMCID: PMC10717155
- DOI: 10.1007/s12576-017-0557-5
Investigating β-adrenergic-induced cardiac hypertrophy through computational approach: classical and non-classical pathways
Abstract
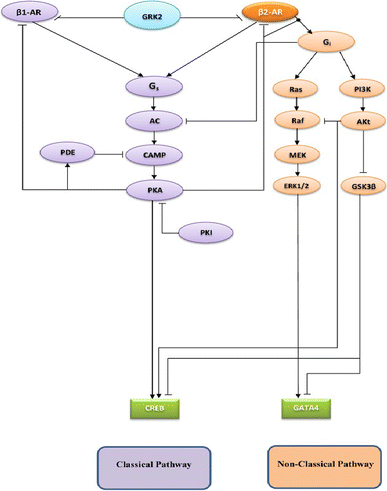
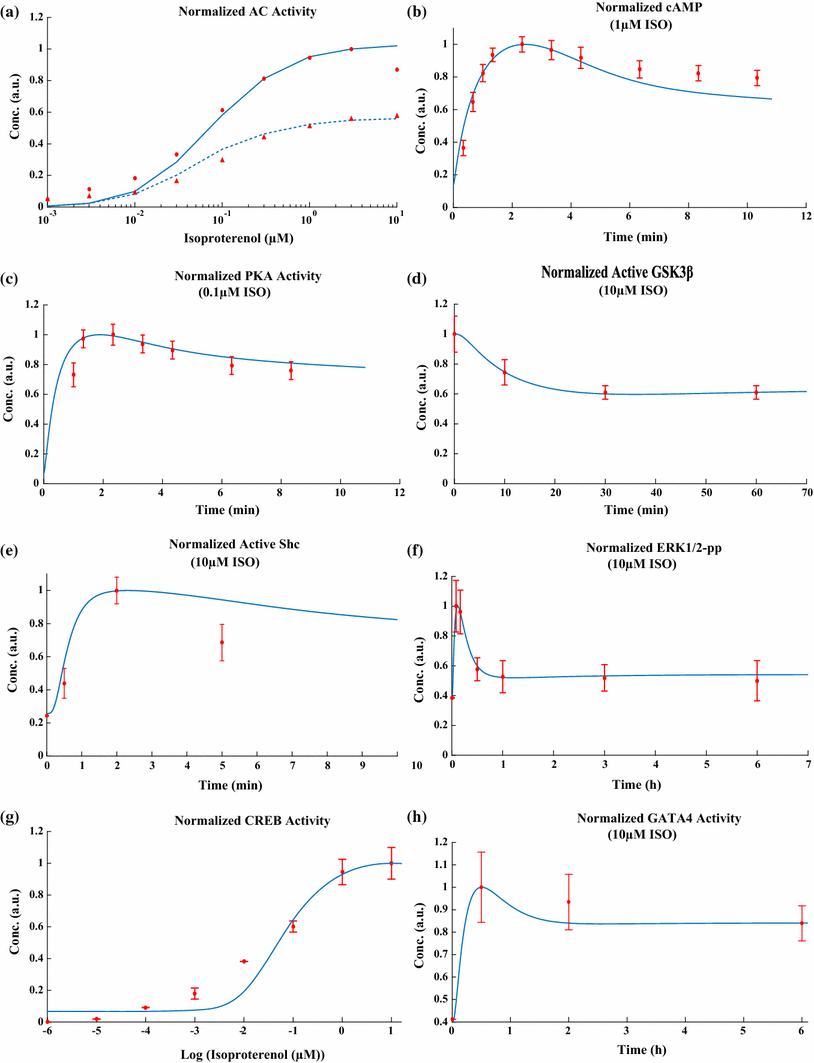
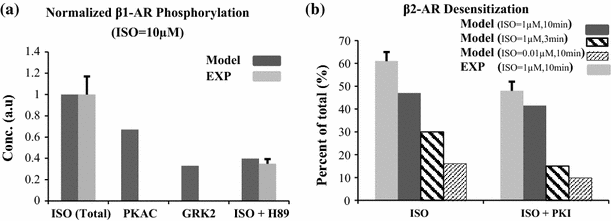
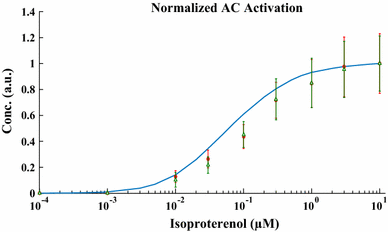
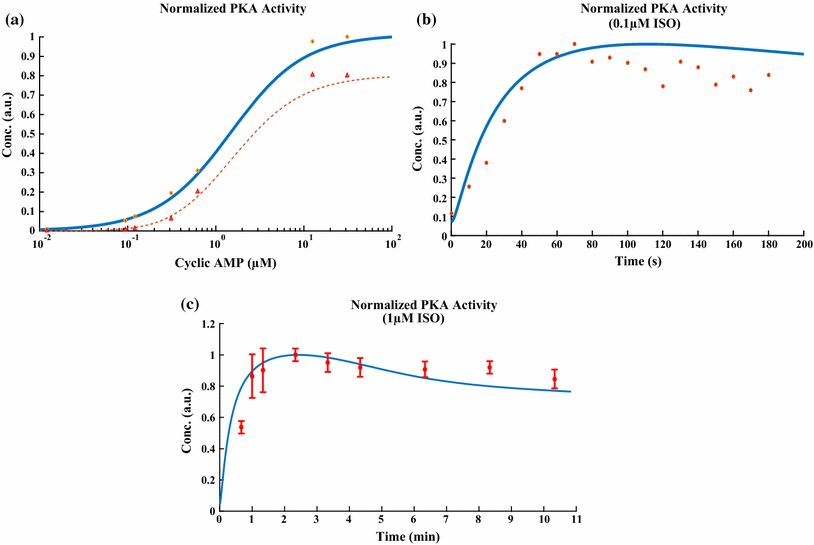
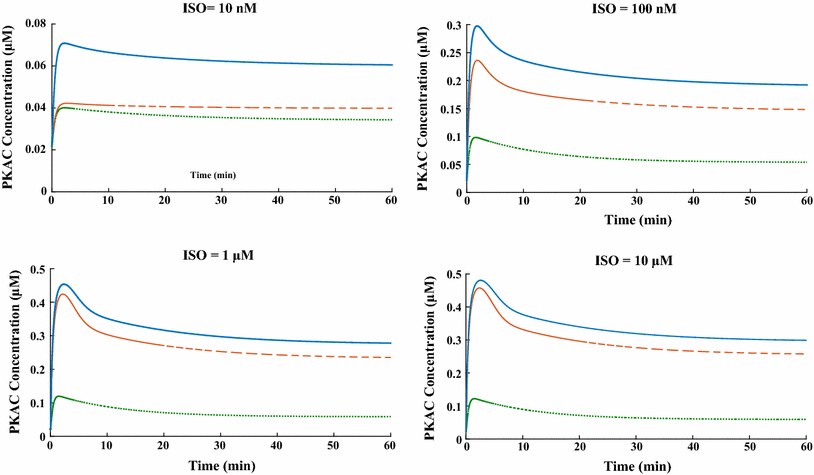
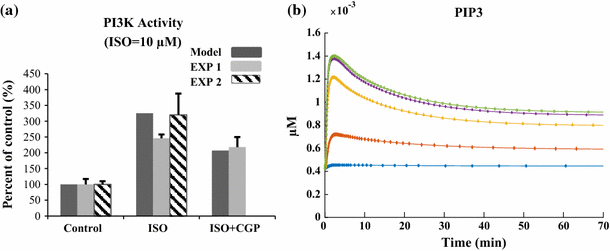
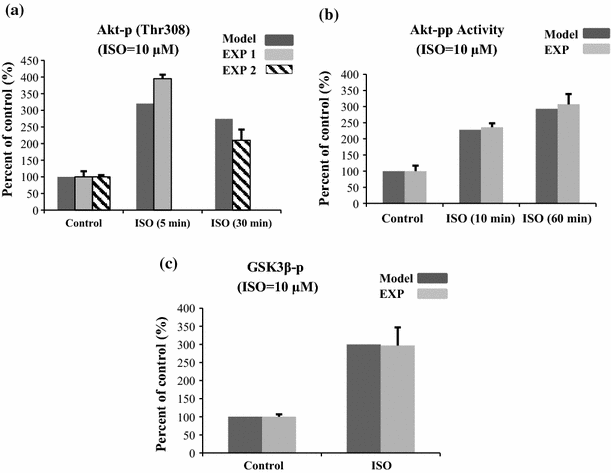
The chronic stimulation of β-adrenergic receptors plays a crucial role in cardiac hypertrophy and its progression to heart failure. In β-adrenergic signaling, in addition to the well-established classical pathway, Gs/AC/cAMP/PKA, activation of non-classical pathways such as Gi/PI3K/Akt/GSK3β and Gi/Ras/Raf/MEK/ERK contribute in cardiac hypertrophy. The signaling network of β-adrenergic-induced hypertrophy is very complex and not fully understood. So, we use a computational approach to investigate the dynamic response and contribution of β-adrenergic mediators in cardiac hypertrophy. The proposed computational model provides insights into the effects of β-adrenergic classical and non-classical pathways on the activity of hypertrophic transcription factors CREB and GATA4. The results illustrate that the model captures the dynamics of the main signaling mediators and reproduces the experimental observations well. The results also show that despite the low portion of β2 receptors out of total cardiac β-adrenergic receptors, their contribution in the activation of hypertrophic mediators and regulation of β-adrenergic-induced hypertrophy is noticeable and variations in β1/β2 receptors ratio greatly affect the ISO-induced hypertrophic response. The model results illustrate that GSK3β deactivation after β-adrenergic receptor stimulation has a major influence on CREB and GATA4 activation and consequent cardiac hypertrophy. Also, it is found through sensitivity analysis that PKB (Akt) activation has both pro-hypertrophic and anti-hypertrophic effects in β-adrenergic signaling.
Keywords: CREB transcription factor; GATA4 transcription factor; Gi/PI3K/Akt/GSK3β pathway; Gi/Ras/Raf/MEK/ERK pathway; Non-classical pathways; β-Adrenergic signaling.
Figures

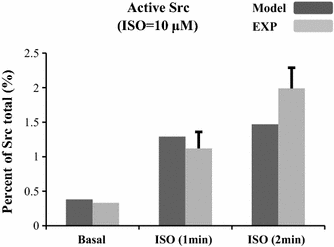
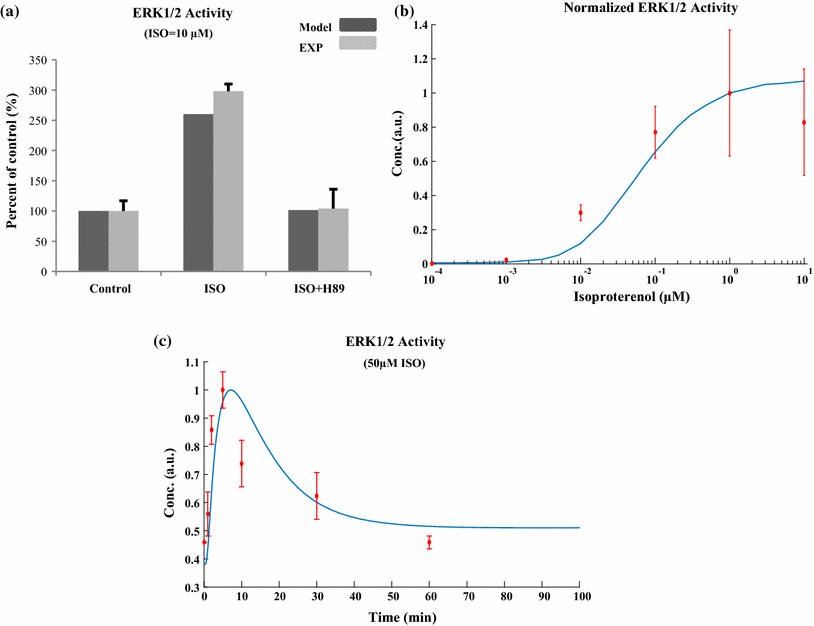
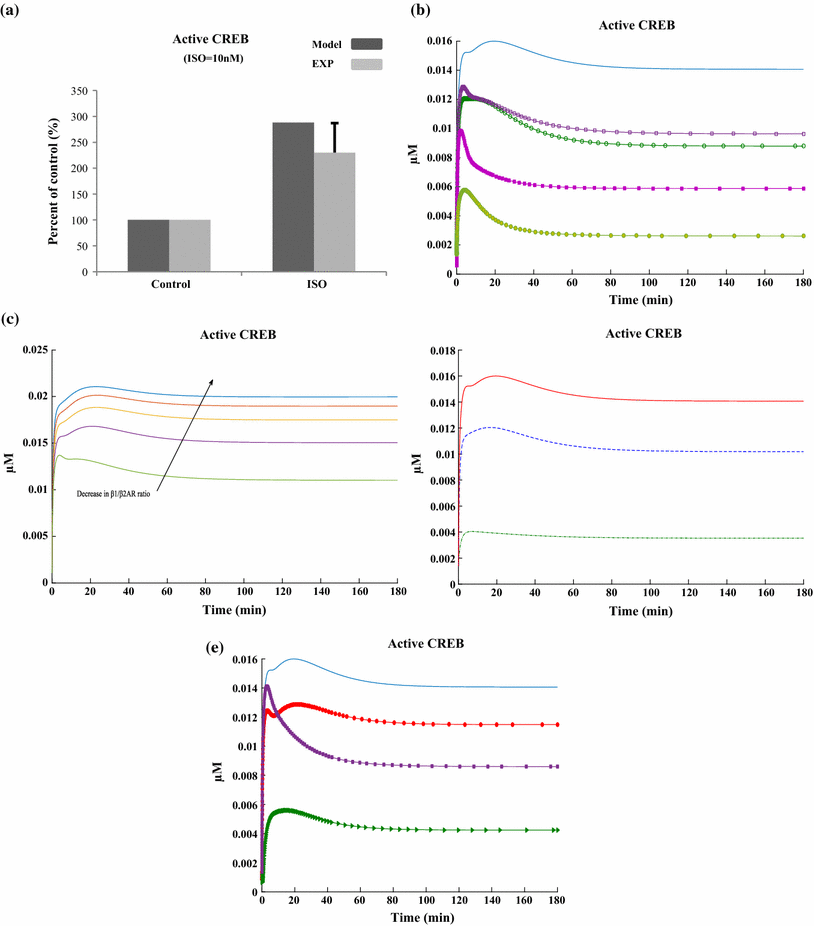


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

