Aberrant Sodium Channel Currents and Hyperexcitability of Medial Entorhinal Cortex Neurons in a Mouse Model of SCN8A Encephalopathy
- PMID: 28676574
- PMCID: PMC5551062
- DOI: 10.1523/JNEUROSCI.2709-16.2017
Aberrant Sodium Channel Currents and Hyperexcitability of Medial Entorhinal Cortex Neurons in a Mouse Model of SCN8A Encephalopathy
Abstract
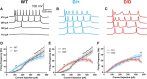
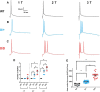
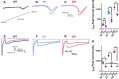
SCN8A encephalopathy, or early infantile epileptic encephalopathy 13 (EIEE13), is caused predominantly by de novo gain-of-function mutations in the voltage-gated Na channel Nav1.6. Affected individuals suffer from refractory seizures, developmental delay, cognitive disability, and elevated risk of sudden unexpected death in epilepsy (SUDEP). A knock-in mouse model carrying the patient mutation p.Asn1768Asp (N1768D) reproduces many features of the disorder, including spontaneous seizures and SUDEP. We used the mouse model to examine the effects of the mutation on layer II stellate neurons of the medial entorhinal cortex (mEC), which transmit excitatory input to the hippocampus. Heterozygous (Scn8aD/+), homozygous (Scn8aD/D)), and WT (Scn8a+/+) littermates were compared at 3 weeks of age, the time of seizure onset for homozygous mice. Heterozygotes remain seizure free for another month. mEC layer II neurons of heterozygous and homozygous mice were hyperexcitable and generated long-lasting depolarizing potentials with bursts of action potentials after synaptic stimulation. Recording of Na currents revealed proexcitatory increases in persistent and resurgent currents and rightward shifts in inactivation parameters, leading to significant increases in the magnitude of window currents. The proexcitatory changes were more pronounced in homozygous mice than in heterozygotes, consistent with the earlier age of seizure onset in homozygotes. These studies demonstrate that the N1768D mutation increases the excitability of mEC layer II neurons by increasing persistent and resurgent Na currents and disrupting channel inactivation. The aberrant activities of mEC layer II neurons would provide excessive excitatory input to the hippocampus and contribute to hyperexcitability of hippocampal neurons in this model of SCN8A encephalopathy.SIGNIFICANCE STATEMENTSCN8A encephalopathy is a devastating neurological disorder that results from de novo mutations in the Na channel Nav1.6. In addition to seizures, patients suffer from cognitive and developmental delays and are at high risk for sudden unexpected death in epilepsy (SUDEP). A mouse knock-in model expressing the patient mutation N1768D reproduces several pathological phenotypes, including spontaneous seizures and sudden death. We demonstrate that medial entorhinal cortex (mEC) neurons from the mouse model exhibit proexcitatory alterations in Na channel activity, some of which were not seen in hippocampal or cortical neurons, and resulting in neuronal hyperexcitability. Because mEC neurons regulate the activity of the hippocampus, which plays an important role in seizure onset, we propose that these profound changes in mEC neuron excitability associated with the gain-of-function mutation of Nav1.6 may increase excitatory drive into the hippocampus, culminating in seizure activity and SUDEP.
Keywords: SCN8A; action potential; entorhinal cortex; epilepsy; epileptic encephalopathy; sodium channels.
Copyright © 2017 the authors 0270-6474/17/377643-13$15.00/0.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
