Kinetic processivity of the two-step oxidations of progesterone and pregnenolone to androgens by human cytochrome P450 17A1
- PMID: 28684414
- PMCID: PMC5555180
- DOI: 10.1074/jbc.M117.794917
Kinetic processivity of the two-step oxidations of progesterone and pregnenolone to androgens by human cytochrome P450 17A1
Abstract
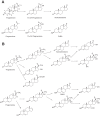
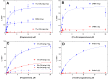
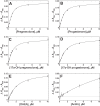
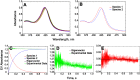
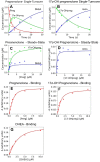
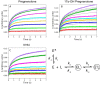
Cytochrome P450 (P450, CYP) 17A1 plays a critical role in steroid metabolism, catalyzing both the 17α-hydroxylation of pregnenolone and progesterone and the subsequent 17α,20-lyase reactions to form dehydroepiandrosterone (DHEA) and androstenedione (Andro), respectively, critical for generating glucocorticoids and androgens. Human P450 17A1 reaction rates examined are enhanced by the accessory protein cytochrome b5 (b5), but the exact role of b5 in P450 17A1-catalyzed reactions is unclear as are several details of these reactions. Here, we examined in detail the processivity of the 17α-hydroxylation and lyase steps. b5 did not enhance reaction rates by decreasing the koff rates of any of the steroids. Steroid binding to P450 17A1 was more complex than a simple two-state system. Pre-steady-state experiments indicated lag phases for Andro production from progesterone and for DHEA from pregnenolone, indicating a distributive character of the enzyme. However, we observed processivity in pregnenolone/DHEA pulse-chase experiments. (S)-Orteronel was three times more inhibitory toward the conversion of 17α-hydroxypregnenolone to DHEA than toward the 17α-hydroxylation of pregnenolone. IC50 values for (S)-orteronel were identical for blocking DHEA formation from pregnenolone and for 17α-hydroxylation, suggestive of processivity. Global kinetic modeling helped assign sets of rate constants for individual or groups of reactions, indicating that human P450 17A1 is an inherently distributive enzyme but that some processivity is present, i.e. some of the 17α-OH pregnenolone formed from pregnenolone did not dissociate from P450 17A1 before conversion to DHEA. Our results also suggest multiple conformations of P450 17A1, as previously proposed on the basis of NMR spectroscopy and X-ray crystallography.
Keywords: cytochrome P450; enzyme kinetics; oxidation-reduction (redox); pre-steady-state kinetics; steady-state kinetics; steroid metabolism; steroidogenesis.
© 2017 by The American Society for Biochemistry and Molecular Biology, Inc.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures

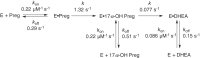
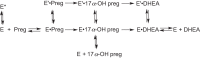
Similar articles
-
Conformational selection dominates binding of steroids to human cytochrome P450 17A1.J Biol Chem. 2019 Jun 28;294(26):10028-10041. doi: 10.1074/jbc.RA119.008860. Epub 2019 May 9. J Biol Chem. 2019. PMID: 31072872 Free PMC article.
-
Structural and kinetic basis of steroid 17α,20-lyase activity in teleost fish cytochrome P450 17A1 and its absence in cytochrome P450 17A2.J Biol Chem. 2015 Feb 6;290(6):3248-68. doi: 10.1074/jbc.M114.627265. Epub 2014 Dec 22. J Biol Chem. 2015. PMID: 25533464 Free PMC article.
-
Similar Rates of Second Electron Transfer and Single-Turnover Dehydroepiandrosterone Formation for Oxyferrous Human Cytochrome P450 17A1 (Steroid 17-Hydroxylase/17,20-lyase)-17-hydroxypregnenolone Complex with Either Human Cytochrome P450-Oxidoreductase or Human Cytochrome b5.Biochemistry. 2025 May 20;64(10):2306-2317. doi: 10.1021/acs.biochem.5c00217. Epub 2025 May 9. Biochemistry. 2025. PMID: 40340376
-
Role of cytochrome b5 in the modulation of the enzymatic activities of cytochrome P450 17α-hydroxylase/17,20-lyase (P450 17A1).J Steroid Biochem Mol Biol. 2017 Jun;170:2-18. doi: 10.1016/j.jsbmb.2016.02.033. Epub 2016 Mar 11. J Steroid Biochem Mol Biol. 2017. PMID: 26976652 Review.
-
Structural insights into the function of steroidogenic cytochrome P450 17A1.Mol Cell Endocrinol. 2017 Feb 5;441:68-75. doi: 10.1016/j.mce.2016.08.035. Epub 2016 Aug 24. Mol Cell Endocrinol. 2017. PMID: 27566228 Free PMC article. Review.
Cited by
-
Tight binding of cytochrome b5 to cytochrome P450 17A1 is a critical feature of stimulation of C21 steroid lyase activity and androgen synthesis.J Biol Chem. 2021 Jan-Jun;296:100571. doi: 10.1016/j.jbc.2021.100571. Epub 2021 Mar 20. J Biol Chem. 2021. PMID: 33753170 Free PMC article.
-
Kinetics of Intermediate Release Enhances P450 11B2-Catalyzed Aldosterone Synthesis.Biochemistry. 2024 Apr 16;63(8):1026-1037. doi: 10.1021/acs.biochem.3c00725. Epub 2024 Apr 2. Biochemistry. 2024. PMID: 38564530 Free PMC article.
-
Steroidogenic cytochrome P450 17A1 structure and function.Mol Cell Endocrinol. 2021 May 15;528:111261. doi: 10.1016/j.mce.2021.111261. Epub 2021 Mar 26. Mol Cell Endocrinol. 2021. PMID: 33781841 Free PMC article. Review.
-
Kinetic Deuterium Isotope Effects in Cytochrome P450 Reactions.Methods Enzymol. 2017;596:217-238. doi: 10.1016/bs.mie.2017.06.036. Epub 2017 Jul 18. Methods Enzymol. 2017. PMID: 28911772 Free PMC article.
-
Formation and Cleavage of C-C Bonds by Enzymatic Oxidation-Reduction Reactions.Chem Rev. 2018 Jul 25;118(14):6573-6655. doi: 10.1021/acs.chemrev.8b00031. Epub 2018 Jun 22. Chem Rev. 2018. PMID: 29932643 Free PMC article. Review.
References
-
- Auchus R. J., and Miller W. L. (2015) in Cytochrome P450: Structure, Mechanism, and Biochemistry (Ortiz de Montellano P. R., ed) 4th Ed., pp. 851–879, Springer, New York
-
- Guengerich F. P. (2015) in Cytochrome P450: Structure, Mechanism, and Biochemistry (Ortiz de Montellano P. R., ed) 4th Ed., pp. 523–785, Springer, New York
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

