In Vivo Indicators of Cytoplasmic, Vacuolar, and Extracellular pH Using pHluorin2 in Candida albicans
- PMID: 28685162
- PMCID: PMC5497024
- DOI: 10.1128/mSphere.00276-17
In Vivo Indicators of Cytoplasmic, Vacuolar, and Extracellular pH Using pHluorin2 in Candida albicans
Abstract
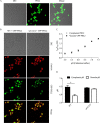
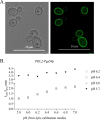
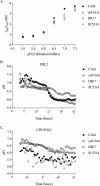
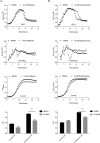
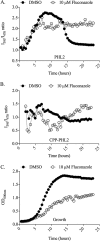
Environmental or chemically induced stresses often trigger physiological responses that regulate intracellular pH. As such, the capacity to detect pH changes in real time and within live cells is of fundamental importance to essentially all aspects of biology. In this respect, pHluorin, a pH-sensitive variant of green fluorescent protein, has provided an invaluable tool to detect such responses. Here, we report the adaptation of pHluorin2 (PHL2), a substantially brighter variant of pHluorin, for use with the human fungal pathogen Candida albicans. As well as a cytoplasmic PHL2 indicator, we describe a version that specifically localizes within the fungal vacuole, an acidified subcellular compartment with important functions in nutrient storage and pH homeostasis. In addition, by means of a glycophosphatidylinositol-anchored PHL2-fusion protein, we generated a cell surface pH sensor. We demonstrated the utility of these tools in several applications, including accurate intracellular and extracellular pH measurements in individual cells via flow cytometry and in cell populations via a convenient plate reader-based protocol. The PHL2 tools can also be used for endpoint as well as time course experiments and to conduct chemical screens to identify drugs that alter normal pH homeostasis. These tools enable observation of the highly dynamic intracellular pH shifts that occur throughout the fungal growth cycle, as well as in response to various chemical treatments. IMPORTANCECandida albicans is an opportunistic fungal pathogen that colonizes the reproductive and gastrointestinal tracts of its human host. It can also invade the bloodstream and deeper organs of immunosuppressed individuals, and thus it encounters enormous variations in external pH in vivo. Accordingly, survival within such diverse niches necessitates robust adaptive responses to regulate intracellular pH. However, the impact of antifungal drugs upon these adaptive responses, and on intracellular pH in general, is not well characterized. Furthermore, the tools and methods currently available to directly monitor intracellular pH in C. albicans, as well as other fungal pathogens, have significant limitations. To address these issues, we developed a new and improved set of pH sensors based on the pH-responsive fluorescent protein pHluorin. This includes a cytoplasmic sensor, a probe that localizes inside the fungal vacuole (an acidified compartment that plays a central role in intracellular pH homeostasis), and a cell surface probe that can detect changes in extracellular pH. These tools can be used to monitor pH within single C. albicans cells or in cell populations in real time through convenient and high-throughput assays.
Keywords: Candida albicans; chemical screening; pH dynamics; vacuoles.
Figures

Similar articles
-
Candida and candidaemia. Susceptibility and epidemiology.Dan Med J. 2013 Nov;60(11):B4698. Dan Med J. 2013. PMID: 24192246 Review.
-
Identification of small molecules that disrupt vacuolar function in the pathogen Candida albicans.PLoS One. 2017 Feb 2;12(2):e0171145. doi: 10.1371/journal.pone.0171145. eCollection 2017. PLoS One. 2017. PMID: 28151949 Free PMC article.
-
Transcriptional profiling in Candida albicans reveals new adaptive responses to extracellular pH and functions for Rim101p.Mol Microbiol. 2004 Dec;54(5):1335-51. doi: 10.1111/j.1365-2958.2004.04350.x. Mol Microbiol. 2004. PMID: 15554973
-
Autophagy in Candida albicans.Methods Enzymol. 2008;451:311-22. doi: 10.1016/S0076-6879(08)03221-7. Methods Enzymol. 2008. PMID: 19185729
-
Gene regulation and host adaptation mechanisms in Candida albicans.Int J Med Microbiol. 2001 May;291(2):183-8. doi: 10.1078/1438-4221-00114. Int J Med Microbiol. 2001. PMID: 11437340 Review.
Cited by
-
Accessibility and contribution to glucan masking of natural and genetically tagged versions of yeast wall protein 1 of Candida albicans.PLoS One. 2018 Jan 12;13(1):e0191194. doi: 10.1371/journal.pone.0191194. eCollection 2018. PLoS One. 2018. PMID: 29329339 Free PMC article.
-
How alkalinization drives fungal pathogenicity.PLoS Pathog. 2017 Nov 9;13(11):e1006621. doi: 10.1371/journal.ppat.1006621. eCollection 2017 Nov. PLoS Pathog. 2017. PMID: 29121119 Free PMC article. Review. No abstract available.
-
Photochromic Fluorophores Enable Imaging of Lowly Expressed Proteins in the Autofluorescent Fungus Candida albicans.mSphere. 2021 Mar 17;6(2):10.1128/msphere.00146-21. doi: 10.1128/mSphere.00146-21. mSphere. 2021. PMID: 33731469 Free PMC article.
-
Candida albicans Hyphal Morphogenesis within Macrophages Does Not Require Carbon Dioxide or pH-Sensing Pathways.Infect Immun. 2023 May 16;91(5):e0008723. doi: 10.1128/iai.00087-23. Epub 2023 Apr 20. Infect Immun. 2023. PMID: 37078861 Free PMC article.
-
Fluorescent toys 'n' tools lighting the way in fungal research.FEMS Microbiol Rev. 2021 Sep 8;45(5):fuab013. doi: 10.1093/femsre/fuab013. FEMS Microbiol Rev. 2021. PMID: 33595628 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
