Effects of Metformin and Furosemide on Rosuvastatin Pharmacokinetics in Healthy Volunteers: Implications for Their Use as Probe Drugs in a Transporter Cocktail
- PMID: 28685495
- PMCID: PMC5794840
- DOI: 10.1007/s13318-017-0427-9
Effects of Metformin and Furosemide on Rosuvastatin Pharmacokinetics in Healthy Volunteers: Implications for Their Use as Probe Drugs in a Transporter Cocktail
Abstract
Background: In a recently described probe drug cocktail for clinically relevant drug transporters containing digoxin, furosemide, metformin and rosuvastatin, mutual interactions were essentially absent except for increases in the systemic exposure of rosuvastatin. To optimize the cocktail, we further examined the dose dependence of the effects of metformin and furosemide on rosuvastatin pharmacokinetics.
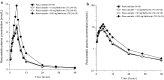
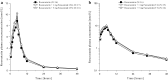
Methods: This was a randomized, open label, single center, six-treatment, six-period, six-sequence crossover trial. Eighteen healthy male subjects received 10 mg rosuvastatin as reference treatment and, as test treatments, 10 mg rosuvastatin combined with 10, 50 or 500 mg metformin (T1, T2 and T3) or with 1 or 5 mg furosemide (T4 and T5). Primary pharmacokinetic endpoints were rosuvastatin C max (maximum plasma concentration) and AUC0-tz (area under the plasma concentration-time curve from time zero to the last quantifiable concentration).
Results: The relative bioavailability of rosuvastatin was essentially unchanged when administered with metformin in T1 and T2, but in T3 it increased to 152% for AUC0-tz (90% CI 135-171%) and 154% for C max (90% CI 132-180%). Coadministration with furosemide did not change rosuvastatin relative bioavailability in T4, but in T5 it increased slightly to 116% for AUC0-tz (90% CI 102-132%) and 118% for C max (90% CI 98-142%).
Conclusion: The increased systemic exposure of rosuvastatin when administered as part of the proposed transporter cocktail is most likely attributable to metformin and only to a minor degree to furosemide. Reduction of the doses of metformin and furosemide is expected to eliminate the previously described interaction. EudraCT no. 2015-003052-46, ClinicalTrials.gov identifier NCT02574845.
Conflict of interest statement
Funding
The study was funded by Boehringer Ingelheim Pharma GmbH & Co. KG, the Sponsor of the trial.
Conflicts of interest
Kathrin Hohl was contracted by Boehringer Ingelheim as an external statistician. All other authors are employees of Boehringer Ingelheim.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The clinical trial protocol was approved by the Ethics Commission of the State Chamber of Physicians of Baden-Württemberg, Stuttgart, Germany, and the Federal Institute for Drugs and Medicinal Products (BfArM), Bonn, Germany.
Informed consent
Written informed consent was obtained from all individual participants included in this trial.
Figures
References
-
- EMA-CHMP. Guideline on the investigation of drug interactions: final (CPMP/EWP/560/95/Rev. 1 corr. 2). 2012 Jun. http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guidelin.... Accessed 13 March 2017.
-
- US-FDA. Guidance for industry: drug interaction studies - study design, data analysis, implications for dosing, and labeling recommendations (draft guidance). 2012 Feb. http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformati.... Accessed 13 March 2017.
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

