Cannabinoids disrupt memory encoding by functionally isolating hippocampal CA1 from CA3
- PMID: 28686594
- PMCID: PMC5521875
- DOI: 10.1371/journal.pcbi.1005624
Cannabinoids disrupt memory encoding by functionally isolating hippocampal CA1 from CA3
Abstract
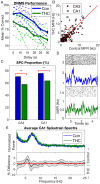
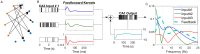
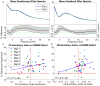
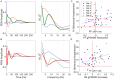
Much of the research on cannabinoids (CBs) has focused on their effects at the molecular and synaptic level. However, the effects of CBs on the dynamics of neural circuits remains poorly understood. This study aims to disentangle the effects of CBs on the functional dynamics of the hippocampal Schaffer collateral synapse by using data-driven nonparametric modeling. Multi-unit activity was recorded from rats doing an working memory task in control sessions and under the influence of exogenously administered tetrahydrocannabinol (THC), the primary CB found in marijuana. It was found that THC left firing rate unaltered and only slightly reduced theta oscillations. Multivariate autoregressive models, estimated from spontaneous spiking activity, were then used to describe the dynamical transformation from CA3 to CA1. They revealed that THC served to functionally isolate CA1 from CA3 by reducing feedforward excitation and theta information flow. The functional isolation was compensated by increased feedback excitation within CA1, thus leading to unaltered firing rates. Finally, both of these effects were shown to be correlated with memory impairments in the working memory task. By elucidating the circuit mechanisms of CBs, these results help close the gap in knowledge between the cellular and behavioral effects of CBs.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
Similar articles
-
Cannabinoids alter spontaneous firing, bursting, and cell synchrony of hippocampal principal cells.Hippocampus. 2011 May;21(5):520-31. doi: 10.1002/hipo.20769. Hippocampus. 2011. PMID: 20101600 Free PMC article.
-
Changes of hippocampal CA3-CA1 population nonlinear dynamics across different training sessions in rats performing a memory-dependent task.Annu Int Conf IEEE Eng Med Biol Soc. 2010;2010:5464-7. doi: 10.1109/IEMBS.2010.5626536. Annu Int Conf IEEE Eng Med Biol Soc. 2010. PMID: 21096285
-
Time Cells in Hippocampal Area CA3.J Neurosci. 2016 Jul 13;36(28):7476-84. doi: 10.1523/JNEUROSCI.0087-16.2016. J Neurosci. 2016. PMID: 27413157 Free PMC article.
-
Acute systemic MK-801 induced functional uncoupling between hippocampal areas CA3 and CA1 with distant effect in the retrosplenial cortex.Hippocampus. 2017 Feb;27(2):134-144. doi: 10.1002/hipo.22678. Epub 2016 Nov 17. Hippocampus. 2017. PMID: 27806441
-
Voltage Imaging in the Study of Hippocampal Circuit Function and Plasticity.Adv Exp Med Biol. 2015;859:197-211. doi: 10.1007/978-3-319-17641-3_8. Adv Exp Med Biol. 2015. PMID: 26238054 Review.
Cited by
-
Nightmares and the Cannabinoids.Curr Neuropharmacol. 2020;18(8):754-768. doi: 10.2174/1570159X18666200114142321. Curr Neuropharmacol. 2020. PMID: 31934840 Free PMC article. Review.
-
Internal representation of hippocampal neuronal population spans a time-distance continuum.Proc Natl Acad Sci U S A. 2019 Apr 9;116(15):7477-7482. doi: 10.1073/pnas.1718518116. Epub 2019 Mar 25. Proc Natl Acad Sci U S A. 2019. PMID: 30910984 Free PMC article.
-
T-DOpE probes reveal sensitivity of hippocampal oscillations to cannabinoids in behaving mice.Nat Commun. 2024 Feb 24;15(1):1686. doi: 10.1038/s41467-024-46021-4. Nat Commun. 2024. PMID: 38402238 Free PMC article.
References
-
- Koppel BS, Brust JC, Fife T, Bronstein J, Youssof S, Gronseth G, et al. Systematic review: Efficacy and safety of medical marijuana in selected neurologic disorders Report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology. 2014;82(17):1556–1563. 10.1212/WNL.0000000000000363 - DOI - PMC - PubMed
-
- Fattore L. Cannabinoids in Neurologic and Mental Disease. Academic Press; 2015.
-
- Blair RE, Deshpande LS, Sombati S, Falenski KW, Martin BR, DeLorenzo RJ. Activation of the cannabinoid type-1 receptor mediates the anticonvulsant properties of cannabinoids in the hippocampal neuronal culture models of acquired epilepsy and status epilepticus. Journal of Pharmacology and Experimental Therapeutics. 2006;317(3):1072–1078. 10.1124/jpet.105.100354 - DOI - PubMed
-
- Deshpande LS, Sombati S, Blair RE, Carter DS, Martin BR, DeLorenzo RJ. Cannabinoid CB1 receptor antagonists cause status epilepticus-like activity in the hippocampal neuronal culture model of acquired epilepsy. Neuroscience letters. 2007;411(1):11–16. 10.1016/j.neulet.2006.09.046 - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

