Plasminogen Tochigi mice exhibit phenotypes similar to wild-type mice under experimental thrombotic conditions
- PMID: 28686706
- PMCID: PMC5501636
- DOI: 10.1371/journal.pone.0180981
Plasminogen Tochigi mice exhibit phenotypes similar to wild-type mice under experimental thrombotic conditions
Abstract
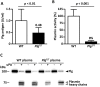
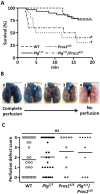
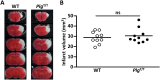
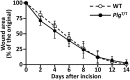
Plasminogen (Plg) is a precursor of plasmin that degrades fibrin. A race-specific A620T mutation in Plg, also known as Plg-Tochigi, originally identified in a patient with recurrent venous thromboembolism, causes dysplasminogenemia with reduced plasmin activity. The Plg-A620T mutation is present in 3-4% of individuals in East Asian populations, and as many as 50,000 Japanese are estimated to be homozygous for the mutant 620T allele. In the present study, to understand the changes of thrombotic phenotypes in individuals with the mutant 620T allele, we generated knock-in mice carrying the homozygous Plg-A622T mutation (PlgT/T), an equivalent to the A620T mutation in human Plg. PlgT/T mice grew normally but showed severely reduced plasmin activity activated by urokinase, equivalent to ~8% of that in wild-type mice. In vitro fibrin clot lysis in plasma was significantly slower in PlgT/T mice than in wild-type mice. However, all experimental models of electrolytic deep vein thrombosis, tissue factor-induced pulmonary embolism, transient focal brain ischaemic stroke, or skin-wound healing showed largely similar phenotypes between PlgT/T mice and wild-type mice. Protein S-K196E mutation (Pros1E/E) is a race-specific genetic risk factor for venous thromboembolism. Coexistence in mice of PlgT/T and Pros1E/E did not affect pulmonary embolism symptoms, compared with those in Pros1E/E mice. Hence, the present study showed that the Plg-A622T mutation, which confers ~8% plasmin activity, does not increase the risk of thrombotic diseases in mice under experimental thrombotic conditions and does not modify the thrombotic phenotype observed in Pros1E/E mice. PlgT/T mice can be used to investigate the potential pathophysiological impact of the Plg-A620T mutation.
Conflict of interest statement
Figures



References
-
- Castellino FJ, Ploplis VA. Structure and function of the plasminogen/plasmin system. Thromb Haemost. 2005;93(4):647–54. doi: 10.1160/TH04-12-0842 . - DOI - PubMed
-
- Schuster V, Hugle B, Tefs K. Plasminogen deficiency. J Thromb Haemost. 2007;5(12):2315–22. doi: 10.1111/j.1538-7836.2007.02776.x . - DOI - PubMed
-
- Rijken DC, Lijnen HR. New insights into the molecular mechanisms of the fibrinolytic system. J Thromb Haemost. 2009;7(1):4–13. doi: 10.1111/j.1538-7836.2008.03220.x . - DOI - PubMed
-
- Schuster V, Mingers AM, Seidenspinner S, Nussgens Z, Pukrop T, Kreth HW. Homozygous mutations in the plasminogen gene of two unrelated girls with ligneous conjunctivitis. Blood. 1997;90(3):958–66. . - PubMed
-
- Tefs K, Gueorguieva M, Klammt J, Allen CM, Aktas D, Anlar FY, et al. Molecular and clinical spectrum of type I plasminogen deficiency: A series of 50 patients. Blood. 2006;108(9):3021–6. doi: 10.1182/blood-2006-04-017350 . - DOI - PubMed
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

