Ivermectin susceptibility and sporontocidal effect in Greater Mekong Subregion Anopheles
- PMID: 28687086
- PMCID: PMC5501099
- DOI: 10.1186/s12936-017-1923-8
Ivermectin susceptibility and sporontocidal effect in Greater Mekong Subregion Anopheles
Abstract
Background: Novel vector control methods that can directly target outdoor malaria transmission are urgently needed in the Greater Mekong Subregion (GMS) to accelerate malaria elimination and artemisinin resistance containment efforts. Ivermectin mass drug administration (MDA) to humans has been shown to effectively kill wild Anopheles and suppress malaria transmission in West Africa. Preliminary laboratory investigations were performed to determine ivermectin susceptibility and sporontocidal effect in GMS Anopheles malaria vectors coupled with pharmacokinetic models of ivermectin at escalating doses.
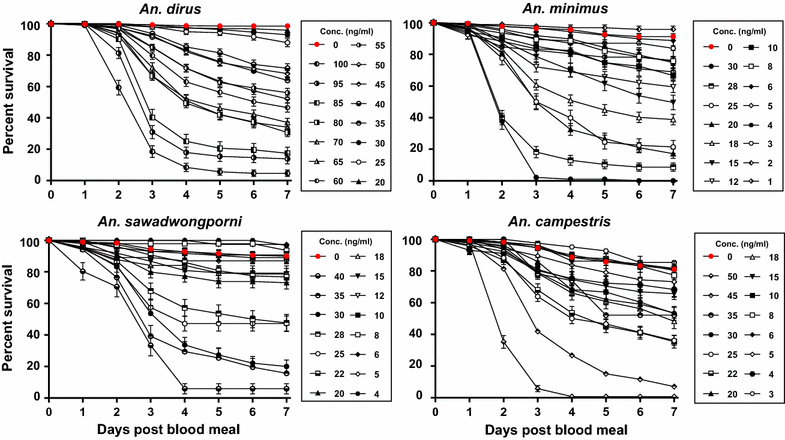
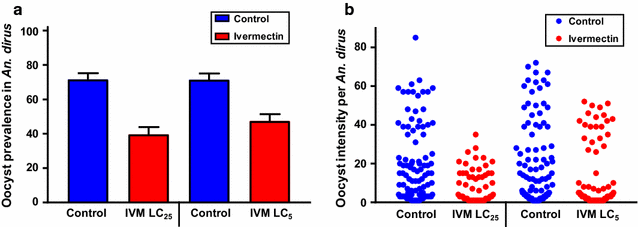
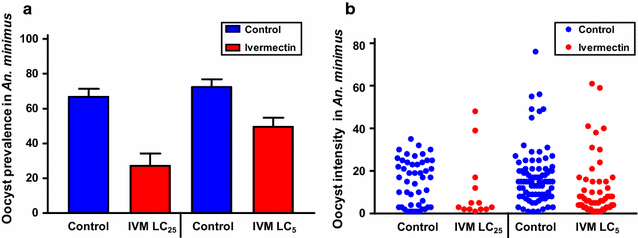
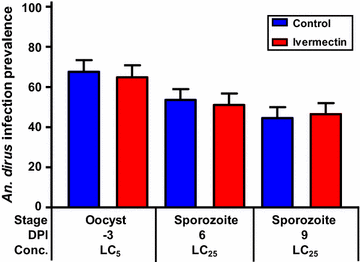
Methods: A population-based pharmacokinetic model of ivermectin was developed using pre-existing data from a clinical trial conducted in Thai volunteers at the 200 µg/kg dose. To assess ivermectin susceptibility, various concentrations of ivermectin compound were mixed in human blood meals and blood-fed to Anopheles dirus, Anopheles minimus, Anopheles sawadwongporni, and Anopheles campestris. Mosquito survival was monitored daily for 7 days and a non-linear mixed effects model with probit analyses was used to calculate concentrations of ivermectin that killed 50% (LC50) of mosquitoes for each species. Blood samples were collected from Plasmodium vivax positive patients and offered to mosquitoes with or without ivermectin at the ivermectin LC25 or LC5 for An. dirus and An. minimus.
Results: The GMS Anopheles displayed a range of susceptibility to ivermectin with species listed from most to least susceptible being An. minimus (LC50 = 16.3 ng/ml) > An. campestris (LC50 = 26.4 ng/ml) = An. sawadwongporni (LC50 = 26.9 ng/ml) > An. dirus (LC50 = 55.6 ng/ml). Mosquito survivorship results, the pharmacokinetic model, and extensive safety data indicated that ivermectin 400 µg/kg is the ideal minimal dose for MDA in the GMS for malaria parasite transmission control. Ivermectin compound was sporontocidal to P. vivax in both An. dirus and An. minimus at the LC25 and LC5 concentrations.
Conclusions: Ivermectin is lethal to dominant GMS Anopheles malaria vectors and inhibits sporogony of P. vivax at safe human relevant concentrations. The data suggest that ivermectin MDA has potential in the GMS as a vector and transmission blocking control tool to aid malaria elimination efforts.
Keywords: Anopheles; Greater Mekong Sub-region; Ivermectin; Plasmodium.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

