Characteristics and expression of genes encoding two small heat shock protein genes lacking introns from Chilo suppressalis
- PMID: 28687981
- PMCID: PMC5741581
- DOI: 10.1007/s12192-017-0823-8
Characteristics and expression of genes encoding two small heat shock protein genes lacking introns from Chilo suppressalis
Abstract
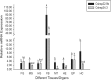
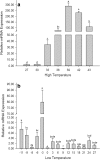
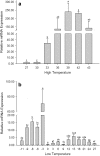
Small heat shock proteins (sHSPs) constitute a large, diverse, and functionally uncharacterized family of heat shock proteins. To gain insight regarding the function of sHSPs in insects, we identified genes encoding two sHSPs, Cshsp22.9b and Cshsp24.3, from the rice pest Chilo suppressalis. The cDNAs of Cshsp22.9b and Cshsp24.3 encoded proteins of 206 and 216 amino acids with isoelectric points of 5.79 and 9.28, respectively. Further characterization indicated that both Cshsp22.9b and Cshsp24.3 lacked introns. Real-time quantitative PCR indicated that Cshsp22.9b and Cshsp24.3 were expressed at higher levels within the fat body as compared to other tissues (head, epidermis, foregut, midgut, hindgut, Malpighian tubules, and hemocytes). Expression of Cshsp22.9b and Cshsp24.3 was lowest in the hindgut and Malpighian tubules, respectively. Cshsp22.9b and Cshsp24.3 showed identical patterns in response to thermal stress from -11 to 43 °C, and both genes were up-regulated by hot and cold temperatures. The mRNA (messenger ribonucleic acid) expression levels of Cshsp22.9b (KY701308) and Cshsp24.3 (KY701309) were highest after a 2-h exposure at 39 °C and started to decline at 42 °C. In response to cold temperatures, both Cshsp22.9b and Cshsp24.3 showed maximal expression after a 2-h exposure to -3 °C. The two Cshsps were more responsive to hot than cold temperature stress and were not induced by mildly cold or warm temperatures. In conclusion, Cshsp22.9b and Cshsp24.3 could play a very important role in the regulation of physiological activities in C. suppressalis that are impacted by environmental stimuli.
Keywords: Chilo suppressalis; Expression; Temperature; Tissues; sHSPs.
Figures



Similar articles
-
Five small heat shock protein genes from Chilo suppressalis: characteristics of gene, genomic organization, structural analysis, and transcription profiles.Cell Stress Chaperones. 2014 Jan;19(1):91-104. doi: 10.1007/s12192-013-0437-8. Epub 2013 May 25. Cell Stress Chaperones. 2014. PMID: 23702967 Free PMC article.
-
Characterization and functional analysis of Cshsp19.0 encoding a small heat shock protein in Chilo suppressalis (Walker).Int J Biol Macromol. 2021 Oct 1;188:924-931. doi: 10.1016/j.ijbiomac.2021.07.186. Epub 2021 Aug 2. Int J Biol Macromol. 2021. PMID: 34352319
-
Chilo suppressalis heat shock proteins are regulated by heat shock factor 1 during heat stress.Insect Mol Biol. 2023 Feb;32(1):69-78. doi: 10.1111/imb.12814. Epub 2022 Nov 1. Insect Mol Biol. 2023. PMID: 36279182
-
Small heat shock proteins: recent developments.Biomol Concepts. 2013 Dec;4(6):583-95. doi: 10.1515/bmc-2013-0028. Biomol Concepts. 2013. PMID: 25436758 Review.
-
The BAG3-dependent and -independent roles of cardiac small heat shock proteins.JCI Insight. 2019 Feb 21;4(4):e126464. doi: 10.1172/jci.insight.126464. eCollection 2019 Feb 21. JCI Insight. 2019. PMID: 30830872 Free PMC article. Review.
Cited by
-
Characterization and Functional Analysis of Small Heat Shock Protein Genes (Hsp22.2 and Hsp26.7) in Sitodiplosis mosellana Diapause.Insects. 2025 Jun 20;16(7):649. doi: 10.3390/insects16070649. Insects. 2025. PMID: 40725281 Free PMC article.
-
Characterization of an inducible HSP70 gene in Chilo suppressalis and expression in response to environmental and biological stress.Cell Stress Chaperones. 2020 Jan;25(1):65-72. doi: 10.1007/s12192-019-01047-2. Epub 2019 Dec 2. Cell Stress Chaperones. 2020. PMID: 31792734 Free PMC article.
-
Transcription dynamics of heat-shock proteins (Hsps) and endosymbiont titres in response to thermal stress in whitefly, Bemisia tabaci (Asia-I).Front Physiol. 2023 Jan 13;13:1097459. doi: 10.3389/fphys.2022.1097459. eCollection 2022. Front Physiol. 2023. PMID: 36714306 Free PMC article.
-
Characterization of genes encoding small heat shock proteins from Bemisia tabaci and expression under thermal stress.PeerJ. 2019 Jun 5;7:e6992. doi: 10.7717/peerj.6992. eCollection 2019. PeerJ. 2019. PMID: 31205823 Free PMC article.
-
Molecular Cloning and Characterization of Small Heat Shock Protein Genes in the Invasive Leaf Miner Fly, Liriomyza trifolii.Genes (Basel). 2019 Oct 3;10(10):775. doi: 10.3390/genes10100775. Genes (Basel). 2019. PMID: 31623413 Free PMC article.
References
-
- Advani NK, Kenkel CD, Davies SW, Parmesan C, Singer MC, Matz MV. Variation in heat shock protein expression at the latitudinal range limits of a widely distributed species, the Glanville fritillary butterfly (Melitaea cinxia) Physiol Entomol. 2016;41(3):241–248. doi: 10.1111/phen.12148. - DOI
-
- Bale JS, Masters GJ, Hodkinson ID, Awmak C, Bezemer TM, Brown V, Butterfield J, Buse A, Coulson JC, Farrar J, Good JEG, Hartley R, Jones TH, Lindroth RL, Press MC, Symrnioudis I, Watt A, Whittaker JB. Herbivory in global climate change research: direct effects of rising temperature on insect herbivores. Glob Chang Biol. 2002;8:1–16. doi: 10.1046/j.1365-2486.2002.00451.x. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

