Latent Cytomegalovirus (CMV) Infection Does Not Detrimentally Alter T Cell Responses in the Healthy Old, But Increased Latent CMV Carriage Is Related to Expanded CMV-Specific T Cells
- PMID: 28694811
- PMCID: PMC5483450
- DOI: 10.3389/fimmu.2017.00733
Latent Cytomegalovirus (CMV) Infection Does Not Detrimentally Alter T Cell Responses in the Healthy Old, But Increased Latent CMV Carriage Is Related to Expanded CMV-Specific T Cells
Abstract
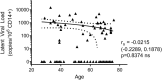
Human cytomegalovirus (HCMV) primary infection and periodic reactivation of latent virus is generally well controlled by T-cell responses in healthy people. In older donors, overt HCMV disease is not generally seen despite the association of HCMV infection with increased risk of mortality. However, increases in HCMV DNA in urine of older people suggest that, although the immune response retains functionality, immunomodulation of the immune response due to lifelong viral carriage may alter its efficacy. Viral transcription is limited during latency to a handful of viral genes and there is both an IFNγ and cellular IL-10 CD4+ T-cell response to HCMV latency-associated proteins. Production of cIL-10 by HCMV-specific CD4+ T-cells is a candidate for aging-related immunomodulation. To address whether long-term carriage of HCMV changes the balance of cIL-10 and IFNγ-secreting T-cell populations, we recruited a large donor cohort aged 23-78 years and correlated T-cell responses to 11 HCMV proteins with age, HCMV IgG levels, latent HCMV load in CD14+ monocytes, and T-cell numbers in the blood. IFNγ responses by CD4+ and CD8+ T-cells to all HCMV proteins were detected, with no age-related increase in this cohort. IL-10-secreting CD4+ T cell responses were predominant to latency-associated proteins but did not increase with age. Quantification of HCMV genomes in CD14+ monocytes, a known site of latent HCMV carriage, did not reveal any increase in viral genome copies in older donors. Importantly, there was a significant positive correlation between the latent viral genome copy number and the breadth and magnitude of the IFNγ T-cell response to HCMV proteins. This study suggests in healthy aged donors that HCMV-specific changes in the T cell compartment were not affected by age and were effective, as viremia was a very rare event. Evidence from studies of unwell aged has shown HCMV to be an important comorbidity factor, surveillance of latent HCMV load and low-level viremia in blood and body fluids, alongside typical immunological measures and assessment of the antiviral capacity of the HCMV-specific immune cell function would be informative in determining if antiviral treatment of HCMV replication in the old maybe beneficial.
Keywords: IFNγ production; cIL-10+CD4+ T cells; human cytomegalovirus; human cytomegalovirus-specific T-cells; immunology of aging; latent viral load; viral latency.
Figures







Similar articles
-
HCMV carriage in the elderly diminishes anti-viral functionality of the adaptive immune response resulting in virus replication at peripheral sites.Front Immunol. 2022 Dec 15;13:1083230. doi: 10.3389/fimmu.2022.1083230. eCollection 2022. Front Immunol. 2022. PMID: 36591233 Free PMC article.
-
Human Cytomegalovirus (HCMV)-Specific CD4+ T Cells Are Polyfunctional and Can Respond to HCMV-Infected Dendritic Cells In Vitro.J Virol. 2017 Feb 28;91(6):e02128-16. doi: 10.1128/JVI.02128-16. Print 2017 Mar 15. J Virol. 2017. PMID: 28053099 Free PMC article.
-
Human cytomegalovirus modulates monocyte-mediated innate immune responses during short-term experimental latency in vitro.J Virol. 2014 Aug;88(16):9391-405. doi: 10.1128/JVI.00934-14. Epub 2014 Jun 11. J Virol. 2014. PMID: 24920803 Free PMC article.
-
Generation, maintenance and tissue distribution of T cell responses to human cytomegalovirus in lytic and latent infection.Med Microbiol Immunol. 2019 Aug;208(3-4):375-389. doi: 10.1007/s00430-019-00598-6. Epub 2019 Mar 20. Med Microbiol Immunol. 2019. PMID: 30895366 Free PMC article. Review.
-
The CD4+ T Cell Response to Human Cytomegalovirus in Healthy and Immunocompromised People.Front Cell Infect Microbiol. 2020 May 19;10:202. doi: 10.3389/fcimb.2020.00202. eCollection 2020. Front Cell Infect Microbiol. 2020. PMID: 32509591 Free PMC article. Review.
Cited by
-
Cytomegalovirus Exposure in the Elderly Does Not Reduce CD8 T Cell Repertoire Diversity.J Immunol. 2019 Jan 15;202(2):476-483. doi: 10.4049/jimmunol.1800217. Epub 2018 Dec 12. J Immunol. 2019. PMID: 30541882 Free PMC article.
-
The Transcriptome of Latent Human Cytomegalovirus.J Virol. 2019 May 15;93(11):e00047-19. doi: 10.1128/JVI.00047-19. Print 2019 Jun 1. J Virol. 2019. PMID: 30867313 Free PMC article. Review.
-
Next-Generation Sequencing Analysis of the Human TCRγδ+ T-Cell Repertoire Reveals Shifts in Vγ- and Vδ-Usage in Memory Populations upon Aging.Front Immunol. 2018 Mar 6;9:448. doi: 10.3389/fimmu.2018.00448. eCollection 2018. Front Immunol. 2018. PMID: 29559980 Free PMC article.
-
Cytomegalovirus Latency and Reactivation: An Intricate Interplay With the Host Immune Response.Front Cell Infect Microbiol. 2020 Mar 31;10:130. doi: 10.3389/fcimb.2020.00130. eCollection 2020. Front Cell Infect Microbiol. 2020. PMID: 32296651 Free PMC article. Review.
-
Parameters of the Immune System and Vitamin D Levels in Old Individuals.Front Immunol. 2018 May 24;9:1122. doi: 10.3389/fimmu.2018.01122. eCollection 2018. Front Immunol. 2018. PMID: 29910802 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

