Effects of Coal Fly Ash Particulate Matter on the Antimicrobial Activity of Airway Surface Liquid
- PMID: 28696208
- PMCID: PMC5744695
- DOI: 10.1289/EHP876
Effects of Coal Fly Ash Particulate Matter on the Antimicrobial Activity of Airway Surface Liquid
Abstract
Background: Sustained exposure to ambient particulate matter (PM) is a global cause of mortality. Coal fly ash (CFA) is a byproduct of coal combustion and is a source of anthropogenic PM with worldwide health relevance. The airway epithelia are lined with fluid called airway surface liquid (ASL), which contains antimicrobial proteins and peptides (AMPs). Cationic AMPs bind negatively charged bacteria to exert their antimicrobial activity. PM arriving in the airways could potentially interact with AMPs in the ASL to affect their antimicrobial activity.
Objectives: We hypothesized that PM can interact with ASL AMPs to impair their antimicrobial activity.
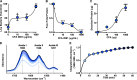
Methods: We exposed pig and human airway explants, pig and human ASL, and the human cationic AMPs β-defensin-3, LL-37, and lysozyme to CFA or control. Thereafter, we assessed the antimicrobial activity of exposed airway samples using both bioluminescence and standard colony-forming unit assays. We investigated PM-AMP electrostatic interaction by attenuated total reflection Fourier-transform infrared spectroscopy and measuring the zeta potential. We also studied the adsorption of AMPs on PM.
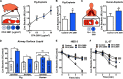
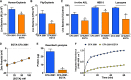
Results: We found increased bacterial survival in CFA-exposed airway explants, ASL, and AMPs. In addition, we report that PM with a negative surface charge can adsorb cationic AMPs and form negative particle-protein complexes.
Conclusion: We propose that when CFA arrives at the airway, it rapidly adsorbs AMPs and creates negative complexes, thereby decreasing the functional amount of AMPs capable of killing pathogens. These results provide a novel translational insight into an early mechanism for how ambient PM increases the susceptibility of the airways to bacterial infection. https://doi.org/10.1289/EHP876.
Figures


References
-
- Ataniiazova RA. 2008. Impact of atmospheric pollution on preschool children's health [in Russian]. Gig Sanit (2):87–89. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
