Adverse Effects and Choice between the Injectable Agents Amikacin and Capreomycin in Multidrug-Resistant Tuberculosis
- PMID: 28696239
- PMCID: PMC5571306
- DOI: 10.1128/AAC.02586-16
Adverse Effects and Choice between the Injectable Agents Amikacin and Capreomycin in Multidrug-Resistant Tuberculosis
Abstract
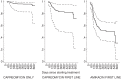
The prolonged use of injectable agents in a regimen for the treatment of multidrug-resistant tuberculosis (MDR-TB) is recommended by the World Health Organization, despite its association with ototoxicity and nephrotoxicity. We undertook this study to look at the relative adverse effects of capreomycin and amikacin. We reviewed the case notes of 100 consecutive patients treated at four MDR-TB treatment centers in the United Kingdom. The median total duration of treatment with an injectable agent was 178 days (interquartile range [IQR], 109 to 192 days; n = 73) for those with MDR-TB, 179 days (IQR, 104 to 192 days; n = 12) for those with MDR-TB plus fluoroquinolone resistance, and 558 days (IQR, 324 to 735 days; n = 8) for those with extensively drug-resistant tuberculosis (XDR-TB). Injectable use was longer for those started with capreomycin (183 days; IQR, 123 to 197 days) than those started with amikacin (119 days; IQR, 83 to 177 days) (P = 0.002). Excluding patients with XDR-TB, 51 of 85 (60%) patients were treated with an injectable for over 6 months and 12 of 85 (14%) were treated with an injectable for over 8 months. Forty percent of all patients discontinued the injectable due to hearing loss. Fifty-five percent of patients experienced ototoxicity, which was 5 times (hazard ratio [HR], 5.2; 95% confidence interval [CI], 1.2 to 22.6; P = 0.03) more likely to occur in those started on amikacin than in those treated with capreomycin only. Amikacin was associated with less hypokalemia than capreomycin (odds ratio, 0.28; 95% CI, 0.11 to 0.72), with 5 of 37 (14%) patients stopping capreomycin due to recurrent electrolyte loss. There was no difference in the number of patients experiencing a rise in the creatinine level of >1.5 times the baseline level. Hearing loss is frequent in this cohort, though its incidence is significantly lower in those starting capreomycin, which should be given greater consideration as a first-line agent.
Keywords: antibiotic resistance; antimicrobial safety; tuberculosis.
Copyright © 2017 American Society for Microbiology.
Figures
References
-
- World Health Organization. 2008. Guidelines for the programmatic management of drug-resistant tuberculosis. World Health Organization, Geneva, Switzerland.
-
- World Health Organization. 2011. Guidelines for the programmatic management of drug-resistant tuberculosis—2011 update. World Health Organization, Geneva, Switzerland. - PubMed
-
- World Health Organization. 2016. The WHO treatment guidelines for drug-resistant tuberculosis (2016 update). World Health Organization, Geneva, Switzerland.
-
- Ahuja SD, Ashkin D, Avendano M, Banerjee R, Bauer M, Bayona JN, Becerra MC, Benedetti A, Burgos M, Centis R, Chan ED, Chiang CY, Cox H, D'Ambrosio L, DeRiemer K, Dung NH, Enarson D, Falzon D, Flanagan K, Flood J, Garcia-Garcia ML, Gandhi N, Granich RM, Hollm-Delgado MG, Holtz TH, Iseman MD, Jarlsberg LG, Keshavjee S, Kim HR, Koh WJ, Lancaster J, Lange C, de Lange WC, Leimane V, Leung CC, Li J, Menzies D, Migliori GB, Mishustin SP, Mitnick CD, Narita M, O'Riordan P, Pai M, Palmero D, Park SK, Pasvol G, Peña J, Pérez-Guzmán C, Quelapio MI, Ponce-de-Leon A. 2012. Multidrug resistant pulmonary tuberculosis treatment regimens and patient outcomes: an individual patient data meta-analysis of 9,153 patients. PLoS Med 9:e1001300. doi: 10.1371/journal.pmed.1001300. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

