Development in the number of clinical trial applications in Western Europe from 2007 to 2015: retrospective study of data from national competent authorities
- PMID: 28698332
- PMCID: PMC5734571
- DOI: 10.1136/bmjopen-2016-015579
Development in the number of clinical trial applications in Western Europe from 2007 to 2015: retrospective study of data from national competent authorities
Abstract
Objective: To investigate the development in the number of applications for authorisation of clinical trials of medicines (CTAs) submitted annually to national competent authorities in 10 Western European member states of the European Union from 2007 to 2015.
Design: Registry study.
Setting: Data from national competent authorities.
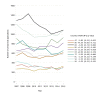
Participants: Germany, Italy, Spain, France, the UK, Belgium, the Netherlands, Austria, Denmark and Sweden. Inclusion criteria were Western European member states of the European Union, receiving more than 200 CTAs per year.
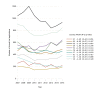
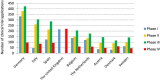
Outcome measures: Summarised number of CTAs and distribution of CTAs by type of sponsor (commercial or non-commercial) and trial phase (I-IV). Average annual growth rates (AAGRs) based on linear regressions. Data were evaluated 2007-2011 and 2012-2015 to compare findings with the European Commission's statement of a 25% decrease in CTAs in the EU from 2007 to 2011.
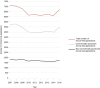
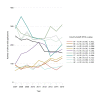
Results: From 2007 to 2011, the summarised number of CTAs decreased significantly (AAGR -3.9% (p=0.02)), primarily due to a decrease in commercially sponsored CTAs. From 2012 to 2015, the change was insignificant (AAGR 2.6% (p=0.27)), however with a 10% increase from 2014 to 2015 after stagnation from 2012 to 2014. Overall, the number of CTAs and distribution by type of sponsor varied considerably between countries. No distinct trends were observed when evaluating CTAs by type of trial phase.
Conclusions: This study found a significant decrease in the number of CTAs in Western Europe from 2007 to 2011 (AAGR -3.9%). This development is possibly attributable to several factors such as the European Clinical Trials Directive, national and local political decisions, and a potential global shift in clinical trial activity. From 2014 to 2015, the number of CTAs increased markedly (10%). However, it is yet too soon to determine if this constitutes a transient fluctuation or a new trend.
Keywords: CLINICAL PHARMACOLOGY; Clinical trials; PUBLIC HEALTH.
© Article author(s) (or their employer(s) unless otherwise stated in the text of the article) 2017. All rights reserved. No commercial use is permitted unless otherwise expressly granted.
Conflict of interest statement
Competing interests: All authors have completed the ICMJE uniform disclosure form at www.icmje.org/coi_disclosure.pdf (available on request from the corresponding author) and declare: no support from any organisation for the submitted work other than those mentioned in the funding statement; MH reports research grants from private companies as stated in the disclosure form; these are not related to the submitted work; no other relationships or activities that could appear to have influenced the submitted work.
Figures
Similar articles
-
Impact assessment of the European Clinical Trials Directive: a longitudinal, prospective, observational study analyzing patterns and trends in clinical drug trial applications submitted since 2001 to regulatory agencies in six EU countries.Trials. 2012 Apr 29;13:53. doi: 10.1186/1745-6215-13-53. Trials. 2012. PMID: 22540886 Free PMC article.
-
Has the UK lost its position as a destination for world-leading clinical research? A comparative analysis of haematological cancer clinical trials performance before Brexit.BMJ Open. 2024 Dec 26;14(12):e086058. doi: 10.1136/bmjopen-2024-086058. BMJ Open. 2024. PMID: 39725437 Free PMC article.
-
Fighting trafficking of falsified and substandard medicinal products in Russia.Int J Risk Saf Med. 2015;27 Suppl 1:S37-40. doi: 10.3233/JRS-150681. Int J Risk Saf Med. 2015. PMID: 26639702
-
Navigating the maze of requirements for obtaining approval of non-interventional studies (NIS) in the European Union.Ger Med Sci. 2015 Nov 17;13:Doc21. doi: 10.3205/000225. eCollection 2015. Ger Med Sci. 2015. PMID: 26633964 Free PMC article. Review.
-
Efaproxiral: GSJ 61, JP 4, KDD 86, RS 4, RSR 13.Drugs R D. 2005;6(3):178-85. doi: 10.2165/00126839-200506030-00007. Drugs R D. 2005. PMID: 15869322 Review.
Cited by
-
Clinical Trial Authorisation: A Final Look Back to Better Appraise the New European Regulation.Rev Recent Clin Trials. 2023;18(3):167-171. doi: 10.2174/1574887118666230320124012. Rev Recent Clin Trials. 2023. PMID: 36959158 Free PMC article.
-
Impact of the Clinical Trials Act 2018 on clinical trial activity in Japan from 2018 to 2020: a retrospective database study using new and conventional Japanese registries.BMJ Open. 2022 Jul 18;12(7):e059092. doi: 10.1136/bmjopen-2021-059092. BMJ Open. 2022. PMID: 35851007 Free PMC article.
-
Trends and variation in data quality and availability on the European Union Clinical Trials Register: A cross-sectional study.Clin Trials. 2022 Apr;19(2):172-183. doi: 10.1177/17407745211073483. Epub 2022 Feb 11. Clin Trials. 2022. PMID: 35144496 Free PMC article.
References
-
- European Medicines Agency. Clinical trials in human medicines [Internet]. 2016. http://www.ema.europa.eu/ema/index.jsp?curl=pages/special_topics/general... (accessed 18 Aug 2016).
-
- European Commission. Proposal for a Regulation of the European Parliament and of the Council on Clinical Trials on Medicinal Products for Human Use, and Repealing Directive 2001/20/EC. Brussels: European Commission, 2012.
-
- Hartmann M. Impact assessment of the European clinical trials directive: a longitudinal, prospective, observational study analyzing patterns and trends in clinical drug trial applications submitted since 2001 to regulatory agencies in six EU countries. Trials 2012;13:53 10.1186/1745-6215-13-53 - DOI - PMC - PubMed
-
- Bengtström M, Nybond L. Clinical trials in the Nordic and Baltic countries: ScanBalt [Internet]. http://www.scanbalt.org/press/news_archive/view?id=2894 (accessed 15 Dec 2015).
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
