Multimodal imaging quality control of epithelia regenerated with cultured human donor corneal limbal epithelial stem cells
- PMID: 28698576
- PMCID: PMC5506064
- DOI: 10.1038/s41598-017-05486-8
Multimodal imaging quality control of epithelia regenerated with cultured human donor corneal limbal epithelial stem cells
Abstract
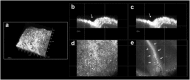
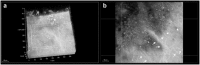
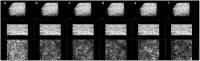
Current imaging techniques for the characterization of differentiated corneal limbal stem cells are destructive and cannot be used in eye bank for monitoring the regenerated epithelium in culture. We presented a minimally invasive, multimodal, marker-free imaging method for the investigation of epithelia regenerated with cultured human donor corneal limbal epithelial stem cells. Two-photon fluorescence and harmonic generation signals were collected from specimens in culture and used for evaluating the structure and morphology of epithelia cultured on two different bio-scaffolds; in addition, donor human corneal tissues were used as controls. The method provided reliable information on the organization of cellular and extracellular components of biomaterial substrates and was highly sensitive to determine differences between the density packing arrangement of epithelial cells of different biomaterials without relying on inferences from exogenous labels. The present minimally invasive standardized quality control methodology can be reliably translated to eye banks and used for monitoring harvested corneal limbal stem cells growth and differentiation in bioengineered materials.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures



Similar articles
-
Comparison of stem cell properties in cell populations isolated from human central and limbal corneal epithelium.Cornea. 2011 Oct;30(10):1155-62. doi: 10.1097/ICO.0b013e318213796b. Cornea. 2011. PMID: 21849892
-
Human corneal epithelial subpopulations: oxygen dependent ex vivo expansion and transcriptional profiling.Acta Ophthalmol. 2013 Jun;91 Thesis 4:1-34. doi: 10.1111/aos.12157. Acta Ophthalmol. 2013. PMID: 23732018
-
The potential for eye bank limbal rings to generate cultured corneal epithelial allografts.Cornea. 2001 Jul;20(5):488-94. doi: 10.1097/00003226-200107000-00010. Cornea. 2001. PMID: 11413404
-
Ocular Surface Reconstruction with the Autologous Conjunctival Epithelium and Establishment of a Feeder-Free and Serum-Free Culture System.Cornea. 2018 Nov;37 Suppl 1:S39-S41. doi: 10.1097/ICO.0000000000001729. Cornea. 2018. PMID: 30211749 Review.
-
Characterization, isolation, expansion and clinical therapy of human corneal epithelial stem/progenitor cells.J Stem Cells. 2014;9(2):79-91. J Stem Cells. 2014. PMID: 25158157 Review.
Cited by
-
Distribution of 50-layer corneal densitometry values and related factors.Int Ophthalmol. 2023 Sep;43(9):3165-3173. doi: 10.1007/s10792-023-02716-z. Epub 2023 Apr 28. Int Ophthalmol. 2023. PMID: 37115476
-
In vivo two-photon microscopy of the human eye.Sci Rep. 2019 Jul 12;9(1):10121. doi: 10.1038/s41598-019-46568-z. Sci Rep. 2019. PMID: 31300680 Free PMC article.
-
Quantitative Discrimination of Healthy and Diseased Corneas With Second Harmonic Generation Microscopy.Transl Vis Sci Technol. 2019 Jun 27;8(3):51. doi: 10.1167/tvst.8.3.51. eCollection 2019 May. Transl Vis Sci Technol. 2019. PMID: 31293806 Free PMC article.
-
Quantitative Analysis of the Corneal Collagen Distribution after In Vivo Cross-Linking with Second Harmonic Microscopy.Biomed Res Int. 2019 Jan 10;2019:3860498. doi: 10.1155/2019/3860498. eCollection 2019. Biomed Res Int. 2019. PMID: 30756083 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

