Identification, Bioactivity, and Productivity of Actinomycins from the Marine-Derived Streptomyces heliomycini
- PMID: 28702007
- PMCID: PMC5487404
- DOI: 10.3389/fmicb.2017.01147
Identification, Bioactivity, and Productivity of Actinomycins from the Marine-Derived Streptomyces heliomycini
Abstract
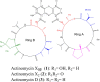
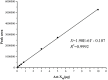
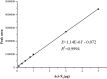
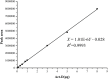
In the process of profiling the secondary metabolites of actinobacteria isolated from the Saudi coastal habitats for production of antibiotics and anti-cancer drugs, the cultures of strain WH1 that was identified as Streptomyces heliomycini exhibited strong antibacterial activity against Staphylococcus aureus. By means of MS and NMR techniques, the active compounds were characterized as actinomycins X0β, X2, and D, respectively. The research on the productivity of this strain for actinomycins revealed that the highest production of actinomycins X0β, X2, and D was reached in the medium MII within 5% salinity and pH 8.5. In this optimized condition, the fermentation titers of actinomycins X0β, X2, and D were 107.6 ± 4.2, 283.4 ± 75.3, and 458.0 ± 76.3 mg/L, respectively. All the three actinomycins X0β, X2, and D showed potent cytotoxicities against the MCF-7, K562, and A549 tumor cell lines, in which actinomycin X2 was the most active against the three tumor cell lines with the IC50 values of 0.8-1.8 nM. Both actinomycins X2 and D showed potent antibacterial activities against S. aureus and the methicillin-resistant S. aureus, Bacillus subtilis, and B. cereus and the actinomycin X2 was more potent.
Keywords: Streptomyces heliomycini WH1; actinomycins; antimicrobial activity; cytotoxicity; marine-derived actinobacteria.
Figures
References
-
- Bian Z.-L., Xiong P.-Y., Shen D.-S. (2003). Determination of bactericidal effect of six antitumor antibiotics on five antibiotic-resistant bacteria. Pharm. Care Res. 3, 253–255. 10.3969/j.issn.1671-2838.2003.04.014 - DOI
-
- Bister B., Bischoff D., Strobele M., Riedlinger J., Reicke A., Wolter F., et al. . (2004). Abyssomicin C–a polycyclic antibiotic from a marine Verrucosispora strain as an inhibitor of the p-aminobenzoic acid/tetrahydrofolate biosynthesis pathway. Angew. Chem. Int. Ed Engl. 43, 2574–2576. 10.1002/anie.200353160 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

