Median raphe region stimulation alone generates remote, but not recent fear memory traces
- PMID: 28708877
- PMCID: PMC5510848
- DOI: 10.1371/journal.pone.0181264
Median raphe region stimulation alone generates remote, but not recent fear memory traces
Abstract
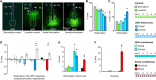
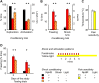
The median raphe region (MRR) is believed to control the fear circuitry indirectly, by influencing the encoding and retrieval of fear memories by amygdala, hippocampus and prefrontal cortex. Here we show that in addition to this established role, MRR stimulation may alone elicit the emergence of remote but not recent fear memories. We substituted electric shocks with optic stimulation of MRR in C57BL/6N male mice in an optogenetic conditioning paradigm and found that stimulations produced agitation, but not fear, during the conditioning trial. Contextual fear, reflected by freezing was not present the next day, but appeared after a 7 days incubation. The optogenetic silencing of MRR during electric shocks ameliorated conditioned fear also seven, but not one day after conditioning. The optogenetic stimulation patterns (50Hz theta burst and 20Hz) used in our tests elicited serotonin release in vitro and lead to activation primarily in the periaqueductal gray examined by c-Fos immunohistochemistry. Earlier studies demonstrated that fear can be induced acutely by stimulation of several subcortical centers, which, however, do not generate persistent fear memories. Here we show that the MRR also elicits fear, but this develops slowly over time, likely by plastic changes induced by the area and its connections. These findings assign a specific role to the MRR in fear learning. Particularly, we suggest that this area is responsible for the durable sensitization of fear circuits towards aversive contexts, and by this, it contributes to the persistence of fear memories. This suggests the existence a bottom-up control of fear circuits by the MRR, which complements the top-down control exerted by the medial prefrontal cortex.
Conflict of interest statement
Figures



References
-
- Johansen JP, Hamanaka H, Monfils MH, Behnia R, Deisseroth K, Blair HT, et al. Optical activation of lateral amygdala pyramidal cells instructs associative fear learning. Proc Natl Acad Sci U S A. 2010;107(28):12692–7. doi: 10.1073/pnas.1002418107 ; PubMed Central PMCID: PMC2906568. - DOI - PMC - PubMed
-
- Bittencourt AS, Carobrez AP, Zamprogno LP, Tufik S, Schenberg LC. Organization of single components of defensive behaviors within distinct columns of periaqueductal gray matter of the rat: role of N-methyl-D-aspartic acid glutamate receptors. Neuroscience. 2004;125(1):71–89. doi: 10.1016/j.neuroscience.2004.01.026 . - DOI - PubMed
-
- Takase LF, Nogueira MI, Baratta M, Bland ST, Watkins LR, Maier SF, et al. Inescapable shock activates serotonergic neurons in all raphe nuclei of rat. Behav Brain Res. 2004;153(1):233–9. doi: 10.1016/j.bbr.2003.12.020 . - DOI - PubMed
-
- Asan E, Steinke M, Lesch KP. Serotonergic innervation of the amygdala: targets, receptors, and implications for stress and anxiety. Histochem Cell Biol. 2013;139(6):785–813. doi: 10.1007/s00418-013-1081-1 . - DOI - PubMed
-
- Sale A, Berardi N, Maffei L. Environment and brain plasticity: towards an endogenous pharmacotherapy. Physiol Rev. 2014;94(1):189–234. doi: 10.1152/physrev.00036.2012 . - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

