A roadmap for evaluating the use and value of durable ventricular assist device therapy
- PMID: 28711453
- PMCID: PMC6746232
- DOI: 10.1016/j.healun.2017.06.008
A roadmap for evaluating the use and value of durable ventricular assist device therapy
Abstract
Heart failure is a disease characterized by profound human suffering with limitations in survival despite treatment with guideline-directed medical therapies. Patients with heart failure frequently progress to advanced stages and often require cardiac transplantation or implantation of left ventricular assist devices (LVADs) to extend survival and improve quality of life. As the number of suitable heart donors, number of experienced medical centers and patient comorbidities place restrictions on the feasibility of cardiac transplantation, implantation of LVADs has emerged as a more frequently applied treatment as either a bridge to transplantation or as permanent therapy. Considerable data have documented improvements in survival, functional status and quality of life offered by LVADs, however, few studies have focused on identifying: (1) determinants of LVAD use across medical centers, (2) the relationship between the determinants of LVAD use and value (defined as quality divided by cost), and (3) how determinants of LVAD use are influenced or impacted by vulnerable populations. We propose a conceptual model that integrates the main determinants of LVAD utilization, which include technology, insurance coverage, market-, provider- and patient-level factors. We propose this paradigm as a necessary prerequisite for understanding LVAD usage and value. This conceptual framework provides a broader view for future studies, which are needed to inform emerging healthcare policies that influence dissemination of this expensive but life-prolonging medical therapy.
Keywords: bridge to transplant; durable LVAD therapy; factors; heart failure; left ventricular assist device.
Copyright © 2018 International Society for the Heart and Lung Transplantation. All rights reserved.
Figures
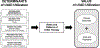

References
-
- Rogers JG, Butler J, Lansman SL, et al.: Chronic mechanical circulatory support for inotrope-dependent heart failure patients who are not transplant candidates: results of the INTrEPID Trial. Journal of the American College of Cardiology 2007;50:741–7. - PubMed
-
- Ho KK, Anderson KM, Kannel WB, Grossman W, Levy D: Survival after the onset of congestive heart failure in Framingham Heart Study subjects. Circulation 1993;88:107. - PubMed
-
- Kirklin JK, Naftel DC, Pagani FD, et al.: Seventh INTERMACS annual report: 15,000 patients and counting. The Journal of heart and lung transplantation : the official publication of the International Society for Heart Transplantation 2015;34:1495–504. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

