Noise-induced cochlear synaptopathy in rhesus monkeys (Macaca mulatta)
- PMID: 28712672
- PMCID: PMC5632522
- DOI: 10.1016/j.heares.2017.07.003
Noise-induced cochlear synaptopathy in rhesus monkeys (Macaca mulatta)
Abstract
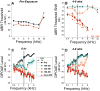
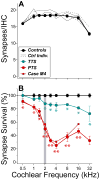
Cochlear synaptopathy can result from various insults, including acoustic trauma, aging, ototoxicity, or chronic conductive hearing loss. For example, moderate noise exposure in mice can destroy up to ∼50% of synapses between auditory nerve fibers (ANFs) and inner hair cells (IHCs) without affecting outer hair cells (OHCs) or thresholds, because the synaptopathy occurs first in high-threshold ANFs. However, the fiber loss likely impairs temporal processing and hearing-in-noise, a classic complaint of those with sensorineural hearing loss. Non-human primates appear to be less vulnerable to noise-induced hair-cell loss than rodents, but their susceptibility to synaptopathy has not been studied. Because establishing a non-human primate model may be important in the development of diagnostics and therapeutics, we examined cochlear innervation and the damaging effects of acoustic overexposure in young adult rhesus macaques. Anesthetized animals were exposed bilaterally to narrow-band noise centered at 2 kHz at various sound-pressure levels for 4 h. Cochlear function was assayed for up to 8 weeks following exposure via auditory brainstem responses (ABRs) and otoacoustic emissions (OAEs). A moderate loss of synaptic connections (mean of 12-27% in the basal half of the cochlea) followed temporary threshold shifts (TTS), despite minimal hair-cell loss. A dramatic loss of synapses (mean of 50-75% in the basal half of the cochlea) was seen on IHCs surviving noise exposures that produced permanent threshold shifts (PTS) and widespread hair-cell loss. Higher noise levels were required to produce PTS in macaques compared to rodents, suggesting that primates are less vulnerable to hair-cell loss. However, the phenomenon of noise-induced cochlear synaptopathy in primates is similar to that seen in rodents.
Keywords: Cochlear histopathology; Cochlear synapatopathy; Noise-induced hearing loss; Non-human primate; Permanent threshold shift; Temporary threshold shift.
Copyright © 2017 Elsevier B.V. All rights reserved.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


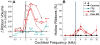

References
-
- Anniko M, Sobin A, Wersall J. Vestibular hair cell pathology in the Shaker-2 mouse. Arch Otorhinolaryngol. 1980;226:45–50. - PubMed
-
- Beyer LA, Odeh H, Probst FJ, Lambert EH, Dolan DF, Camper SA, Kohrman DC, Raphael Y. Hair cells in the inner ear of the pirouette and shaker 2 mutant mice. J Neurocytol. 2000;29:227–39. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

