MUC1-Mediated Metabolic Alterations Regulate Response to Radiotherapy in Pancreatic Cancer
- PMID: 28720669
- PMCID: PMC5626603
- DOI: 10.1158/1078-0432.CCR-17-1151
MUC1-Mediated Metabolic Alterations Regulate Response to Radiotherapy in Pancreatic Cancer
Abstract
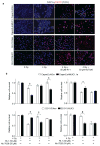
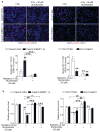
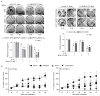
Purpose:MUC1, an oncogene overexpressed in multiple solid tumors, including pancreatic cancer, reduces overall survival and imparts resistance to radiation and chemotherapies. We previously identified that MUC1 facilitates growth-promoting metabolic alterations in pancreatic cancer cells. The present study investigates the role of MUC1-mediated metabolism in radiation resistance of pancreatic cancer by utilizing cell lines and in vivo models.Experimental Design: We used MUC1-knockdown and -overexpressed cell line models for evaluating the role of MUC1-mediated metabolism in radiation resistance through in vitro cytotoxicity, clonogenicity, DNA damage response, and metabolomic evaluations. We also investigated whether inhibition of glycolysis could revert MUC1-mediated metabolic alterations and radiation resistance by using in vitro and in vivo models.Results: MUC1 expression diminished radiation-induced cytotoxicity and DNA damage in pancreatic cancer cells by enhancing glycolysis, pentose phosphate pathway, and nucleotide biosynthesis. Such metabolic reprogramming resulted in high nucleotide pools and radiation resistance in in vitro models. Pretreatment with the glycolysis inhibitor 3-bromopyruvate abrogated MUC1-mediated radiation resistance both in vitro and in vivo, by reducing glucose flux into nucleotide biosynthetic pathways and enhancing DNA damage, which could again be reversed by pretreatment with nucleoside pools.Conclusions: MUC1-mediated nucleotide metabolism plays a key role in facilitating radiation resistance in pancreatic cancer and targeted effectively through glycolytic inhibition. Clin Cancer Res; 23(19); 5881-91. ©2017 AACR.
©2017 American Association for Cancer Research.
Figures



Similar articles
-
MUC1 mucin stabilizes and activates hypoxia-inducible factor 1 alpha to regulate metabolism in pancreatic cancer.Proc Natl Acad Sci U S A. 2012 Aug 21;109(34):13787-92. doi: 10.1073/pnas.1203339109. Epub 2012 Aug 6. Proc Natl Acad Sci U S A. 2012. PMID: 22869720 Free PMC article.
-
Up-Regulation of Non-Homologous End-Joining by MUC1.Genes (Basel). 2024 Jun 19;15(6):808. doi: 10.3390/genes15060808. Genes (Basel). 2024. PMID: 38927743 Free PMC article.
-
A metabolic switch to the pentose-phosphate pathway induces radiation resistance in pancreatic cancer.Radiother Oncol. 2025 Jan;202:110606. doi: 10.1016/j.radonc.2024.110606. Epub 2024 Nov 8. Radiother Oncol. 2025. PMID: 39521275
-
MUC1 oncoprotein mitigates ER stress via CDA-mediated reprogramming of pyrimidine metabolism.Oncogene. 2020 Apr;39(16):3381-3395. doi: 10.1038/s41388-020-1225-4. Epub 2020 Feb 26. Oncogene. 2020. PMID: 32103170 Free PMC article.
-
MUC1: a novel metabolic master regulator.Biochim Biophys Acta. 2014 Apr;1845(2):126-35. doi: 10.1016/j.bbcan.2014.01.001. Epub 2014 Jan 11. Biochim Biophys Acta. 2014. PMID: 24418575 Free PMC article. Review.
Cited by
-
Metabolism of pancreatic cancer: paving the way to better anticancer strategies.Mol Cancer. 2020 Mar 2;19(1):50. doi: 10.1186/s12943-020-01169-7. Mol Cancer. 2020. PMID: 32122374 Free PMC article. Review.
-
Structural Manipulations of Marine Natural Products Inspire a New Library of 3-Amino-1,2,4-Triazine PDK Inhibitors Endowed with Antitumor Activity in Pancreatic Ductal Adenocarcinoma.Mar Drugs. 2023 May 4;21(5):288. doi: 10.3390/md21050288. Mar Drugs. 2023. PMID: 37233482 Free PMC article.
-
Metabolomic profiling of pancreatic adenocarcinoma reveals key features driving clinical outcome and drug resistance.EBioMedicine. 2021 Apr;66:103332. doi: 10.1016/j.ebiom.2021.103332. Epub 2021 Apr 13. EBioMedicine. 2021. PMID: 33862584 Free PMC article.
-
Molecular and Physiological Evaluation of Pancreatic Cancer-Induced Cachexia.Methods Mol Biol. 2019;1882:321-333. doi: 10.1007/978-1-4939-8879-2_28. Methods Mol Biol. 2019. PMID: 30378066 Free PMC article.
-
Development of [225Ac]Ac-DOTA-C595 as radioimmunotherapy of pancreatic cancer: in vitro evaluation, dosimetric assessment and detector calibration.EJNMMI Radiopharm Chem. 2023 Sep 7;8(1):22. doi: 10.1186/s41181-023-00209-z. EJNMMI Radiopharm Chem. 2023. PMID: 37679594 Free PMC article.
References
-
- Roldan GE, Gunderson LL, Nagorney DM, Martin JK, Ilstrup DM, Holbrook MA, et al. External beam versus intraoperative and external beam irradiation for locally advanced pancreatic cancer. Cancer. 1988;61(6):1110–6. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

