Interfaces and ventilator settings for long-term noninvasive ventilation in COPD patients
- PMID: 28721033
- PMCID: PMC5500538
- DOI: 10.2147/COPD.S132170
Interfaces and ventilator settings for long-term noninvasive ventilation in COPD patients
Abstract
Introduction: The establishment of high-intensity (HI) noninvasive ventilation (NIV) that targets elevated PaCO2 has led to an increase in the use of long-term NIV to treat patients with chronic hypercapnic COPD. However, the role of the ventilation interface, especially in more aggressive ventilation strategies, has not been systematically assessed.
Methods: Ventilator settings and NIV compliance were assessed in this prospective cross-sectional monocentric cohort study of COPD patients with pre-existing NIV. Daytime arterialized blood gas analyses and lung function testing were also performed. The primary end point was the distribution among study patients of interfaces (full-face masks [FFMs] vs nasal masks [NMs]) in a real-life setting.
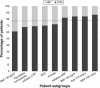
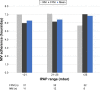
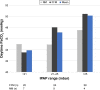
Results: The majority of the 123 patients studied used an FFM (77%), while 23% used an NM. Ventilation settings were as follows: mean ± standard deviation (SD) inspiratory positive airway pressure (IPAP) was 23.2±4.6 mbar and mean ± SD breathing rate was 16.7±2.4/minute. Pressure support ventilation (PSV) mode was used in 52.8% of patients, while assisted pressure-controlled ventilation (aPCV) was used in 47.2% of patients. Higher IPAP levels were associated with an increased use of FFMs (IPAP <21 mbar: 73% vs IPAP >25 mbar: 84%). Mean compliance was 6.5 hours/day, with no differences between FFM (6.4 hours/day) and NM (6.7 hours/day) users. PaCO2 assessment of ventilation quality revealed comparable results among patients with FFMs or NMs.
Conclusion: This real-life trial identified the FFM as the predominantly used interface in COPD patients undergoing long-term NIV. The increased application of FFMs is, therefore, likely to be influenced by higher IPAP levels, which form part of the basis for successful application of HI-NIV in clinical practice.
Keywords: compliance; home mechanical ventilation; interfaces; masks; pressure support; ventilation modes.
Conflict of interest statement
Disclosure JC received speaking fees and travel funding from VitalAire. FSM received speaking/consulting fees from SenTec and Philipps Respironics and travel funding from Vivisol and Heinen und Löwenstein. SBS received speaking/consulting fees from Philipps Respironics and travel funding from SenTec and Heinen und Löwenstein. WW received speaking fees from Heinen und Löwenstein, VitalAire, ResMed, Sentec, Maquet, and Vivisol. JHS received speaking/consulting fees and/or travel funding from Heinen&Löwenstein, VitalAire, ResMed, Linde, Radiometer, Keller Medical, Weinmann, Fisher&Paykel, GHD Gesundheits GmbH, Breas Medical, Respironics, and SenTec AG. MB and ST report no conflicts of interest in this work and JC, FSM, SBS, WW, and JHS report no other conflicts of interest in this work.
Figures


References
-
- Dreher M, Storre JH, Schmoor C, Windisch W. High-intensity versus low-intensity non-invasive ventilation in patients with stable hypercapnic COPD: a randomised crossover trial. Thorax. 2010;65(4):303–308. - PubMed
-
- Köhnlein T, Windisch W, Köhler D, et al. Non-invasive positive pressure ventilation for the treatment of severe stable chronic obstructive pulmonary disease: a prospective, multicentre, randomised, controlled clinical trial. Lancet Respir Med. 2014;2(9):698–705. - PubMed
-
- Struik FM, Sprooten RT, Kerstjens HA, et al. Nocturnal non-invasive ventilation in COPD patients with prolonged hypercapnia after ventilatory support for acute respiratory failure: a randomised, controlled, parallel-group study. Thorax. 2014;69(9):826–834. - PubMed
-
- McEvoy RD, Pierce RJ, Hillman D, et al. Australian trial of non-invasive Ventilation in Chronic Airflow Limitation (AVCAL) Study Group Nocturnal non-invasive nasal ventilation in stable hypercapnic COPD: a randomised controlled trial. Thorax. 2009;64(7):561–566. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

