A Method of Managing Severe Traumatic Brain Injury in the Absence of Intracranial Pressure Monitoring: The Imaging and Clinical Examination Protocol
- PMID: 28726590
- PMCID: PMC5757082
- DOI: 10.1089/neu.2016.4472
A Method of Managing Severe Traumatic Brain Injury in the Absence of Intracranial Pressure Monitoring: The Imaging and Clinical Examination Protocol
Abstract
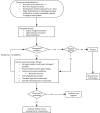
The imaging and clinical examination (ICE) algorithm used in the Benchmark Evidence from South American Trials: Treatment of Intracranial Pressure (BEST TRIP) randomized controlled trial is the only prospectively investigated clinical protocol for traumatic brain injury management without intracranial pressure (ICP) monitoring. As the default literature standard, it warrants careful evaluation. We present the ICE protocol in detail and analyze the demographics, outcome, treatment intensity, frequency of intervention usage, and related adverse events in the ICE-protocol cohort. The 167 ICE protocol patients were young (median 29 years) with a median Glasgow Coma Scale motor score of 4 but with anisocoria or abnormal pupillary reactivity in 40%. This protocol produced outcomes not significantly different from those randomized to the monitor-based protocol (favorable 6-month extended Glasgow Outcome Score in 39%; 41% mortality rate). Agents commonly employed to treat suspected intracranial hypertension included low-/moderate-dose hypertonic saline (72%) and mannitol (57%), mild hyperventilation (adjusted partial pressure of carbon dioxide 30-35 mm Hg in 73%), and pressors to maintain cerebral perfusion (62%). High-dose hyperosmotics or barbiturates were uncommonly used. Adverse event incidence was low and comparable to the BEST TRIP monitored group. Although this protocol should produce similar/acceptable results under circumstances comparable to those in the trial, influences such as longer pre-hospital times and non-specialist transport personnel, plus an intensive care unit model of aggressive physician-intensive care by small groups of neurotrauma-focused intensivists, which differs from most high-resource models, support caution in expecting the same results in dissimilar settings. Finally, this protocol's ICP-titration approach to suspected intracranial hypertension (vs. crisis management for monitored ICP) warrants further study.
Keywords: global health; intracranial hypertension; intracranial pressure monitoring; neurocritical care; severe traumatic brain injury.
Conflict of interest statement
No competing financial interests exist.
Integra LifeSciences donated the ICP monitoring catheters and monitors and provided additional unrestricted support for the parent study. Integra had no role in study design or conduct, data analysis, or the writing of any manuscripts.
Figures

References
-
- Ghajar J., Hariri R.J., Narayan R.K., Iacono L.A., Firlik K., and Patterson R.H. (1995). Survey of critical care management of comatose, head-injured patients in the United States. Crit. Care Med. 23, 560–567 - PubMed
-
- Hesdorffer D.C. and Ghajar J. (2007). Marked improvement in adherence to traumatic brain injury guidelines in United States trauma centers. J. Trauma 63, 841–847 - PubMed
-
- Myburgh J.A., Cooper D.J., Finfer S.R., Venkatesh B., Jones D., Higgins A., Bishop N., and Higlett T. (2008). Epidemiology and 12-month outcomes from traumatic brain injury in Australia and New Zealand. J. Trauma 64, 854–862 - PubMed
-
- Sahjpaul R. and Girotti M. (2000). Intracranial pressure monitoring in severe traumatic brain injury–results of a Canadian survey. Can. J. Neurol. Sci. 27, 143–147 - PubMed
-
- Stocchetti N., Penny K.I., Dearden M., Braakman R., Cohadon F., Iannotti F., Lapierre F., Karimi A., Maas A., Jr., Murray G.D., Ohman J., Persson L., Servadei F., Teasdale G.M., Trojanowski T., and Unterberg A. (2001). Intensive care management of head-injured patients in Europe: a survey from the European brain injury consortium. Intensive Care Med. 27, 400–406 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

