Effects of marine collagen peptides on glucose metabolism and insulin resistance in type 2 diabetic rats
- PMID: 28740282
- PMCID: PMC5502017
- DOI: 10.1007/s13197-017-2663-z
Effects of marine collagen peptides on glucose metabolism and insulin resistance in type 2 diabetic rats
Abstract
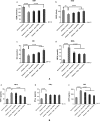
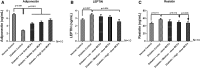
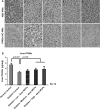
The present study was conducted to investigate the effects of marine collagen peptides (MCPs) on glucose metabolism and insulin resistance using a rat model of type 2 diabetes mellitus (T2DM). Forty T2DM obese Wistar rats were randomly assigned to receive varying doses of MCPs or a vehicle control for 4 weeks. Blood glucose and insulin levels, as well as oxidative stress and inflammation were measured. The expression of glucose transporter type 4 (GLUT4) in skeletal muscles and peroxisome proliferator-activated receptor-α (PPAR-α) in livers of T2DM rats was also measured. It was found that in the group of 9.0 g/kg/day MCPs significantly improved glucose, insulin, and homeostatic model assessment-insulin resistance, and increased the insulin sensitivity index (ISI). In addition, the groups of 4.5 and 2.25 g/kg/day MCPs significantly improved liver steatosis. It was also found that MCPs decreased expression of oxidative stress biomarkers and inflammatory cytokines and adipocytokines in T2DM rats. In conclusion, medium and high doses of MCPs (≥4.5 g/kg/day) improved glucose metabolism and insulin sensitivity in T2DM rats. These beneficial effects of MCPs may be mediated by decreasing oxidative stress and inflammation and by up-regulating GLUT4, and PPAR-α activity.
Keywords: Adipocytokine; Inflammatory cytokines; Insulin resistance; Marine collagen peptide; Oxidative stress.
Conflict of interest statement
The authors have no potential conflicts of interest relevant to this article.
Figures

References
LinkOut - more resources
Full Text Sources
Other Literature Sources
