Shifted dynamic interactions between subcortical nuclei and inferior frontal gyri during response preparation in persistent developmental stuttering
- PMID: 28741037
- PMCID: PMC5772149
- DOI: 10.1007/s00429-017-1476-1
Shifted dynamic interactions between subcortical nuclei and inferior frontal gyri during response preparation in persistent developmental stuttering
Abstract
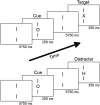
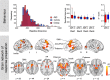
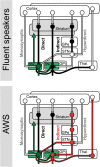
Persistent developmental stuttering is associated with basal ganglia dysfunction or dopamine dysregulation. Here, we studied whole-brain functional connectivity to test how basal ganglia structures coordinate and reorganize sensorimotor brain networks in stuttering. To this end, adults who stutter and fluent speakers (control participants) performed a response anticipation paradigm in the MRI scanner. The preparation of a manual Go/No-Go response reliably produced activity in the basal ganglia and thalamus and particularly in the substantia nigra. Strikingly, in adults who stutter, substantia nigra activity correlated positively with stuttering severity. Furthermore, functional connectivity analyses yielded altered task-related network formations in adults who stutter compared to fluent speakers. Specifically, in adults who stutter, the globus pallidus and the thalamus showed increased network synchronization with the inferior frontal gyrus. This implies dynamic shifts in the response preparation-related network organization through the basal ganglia in the context of a non-speech motor task in stuttering. Here we discuss current findings in the traditional framework of how D1 and D2 receptor activity shapes focused movement selection, thereby suggesting a disproportional involvement of the direct and the indirect pathway in stuttering.
Keywords: Basal ganglia; Disinhibition; Inferior frontal gyrus; Persistent developmental stuttering; Response anticipation; Substantia nigra.
Figures




References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

