Topical moringin-cream relieves neuropathic pain by suppression of inflammatory pathway and voltage-gated ion channels in murine model of multiple sclerosis
- PMID: 28741431
- PMCID: PMC5555508
- DOI: 10.1177/1744806917724318
Topical moringin-cream relieves neuropathic pain by suppression of inflammatory pathway and voltage-gated ion channels in murine model of multiple sclerosis
Abstract
Background: Neuropathic pain represents the major public health burden with a strong impact on quality life in multiple sclerosis patients. Although some advances have been obtained in the last years, the conventional therapies remain poorly effective. Thus, the discovery of innovative approaches to improve the outcomes for multiple sclerosis patients is a goal of primary importance. With this aim, we investigated the efficacy of the 4-(α−L-rhamnopyranosyloxy)benzyl isothiocyanate (moringin), purified from Moringa oleifera seeds and ready-to-use as topical treatment in experimental autoimmune encephalomyelitis, murine model of multiple sclerosis. Female C57BL/6 mice immunized with myelin oligodendrocyte glycoprotein (MOG35–55) were topically treated with 2% moringin cream twice daily from the onset of the symptoms until the sacrifice occurred about 21 days after experimental autoimmune encephalomyelitis induction.
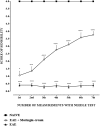
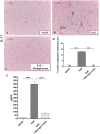
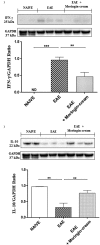
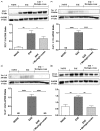
Results: Our observations showed the efficacy of 2% moringin cream treatment in reducing clinical and histological disease score, as well as in alleviating neuropathic pain with consequent recovering of the hind limbs and response to mechanical stimuli. In particular, Western blot analysis and immunohistochemical evaluations revealed that 2% moringin cream was able to counteract the inflammatory cascade by reducing the production of pro-inflammatory cytokines (interleukin-17 and interferon-γ) and in parallel by increasing the expression of anti-inflammatory cytokine (interleukin-10). Interestingly, 2% moringin cream treatment was found to modulate the expression of voltage-gated ion channels (results focused on P2X7, Nav 1.7, Nav 1.8 KV4.2, and α2δ-1) as well as metabotropic glutamate receptors (mGluR5 and xCT) involved in neuropathic pain initiation and maintenance.
Conclusions: Finally, our evidences suggest 2% moringin cream as a new pharmacological trend in the management of multiple sclerosis-induced neuropathic pain.
Figures



Similar articles
-
A new formulation of cannabidiol in cream shows therapeutic effects in a mouse model of experimental autoimmune encephalomyelitis.Daru. 2015 Oct 21;23:48. doi: 10.1186/s40199-015-0131-8. Daru. 2015. PMID: 26489494 Free PMC article.
-
Moringin activates Wnt canonical pathway by inhibiting GSK3β in a mouse model of experimental autoimmune encephalomyelitis.Drug Des Devel Ther. 2016 Oct 4;10:3291-3304. doi: 10.2147/DDDT.S110514. eCollection 2016. Drug Des Devel Ther. 2016. PMID: 27784989 Free PMC article.
-
Antiinflammatory activity of glucomoringin isothiocyanate in a mouse model of experimental autoimmune encephalomyelitis.Fitoterapia. 2014 Jun;95:160-74. doi: 10.1016/j.fitote.2014.03.018. Epub 2014 Mar 29. Fitoterapia. 2014. PMID: 24685508
-
Managing Neuropathic Pain in Multiple Sclerosis: Pharmacological Interventions.Med Chem. 2018 Feb 6;14(2):106-119. doi: 10.2174/1573406413666170906122508. Med Chem. 2018. PMID: 28875858 Review.
-
Injury-Induced Effectors of Neuropathic Pain.Mol Neurobiol. 2020 Jan;57(1):51-66. doi: 10.1007/s12035-019-01756-w. Epub 2019 Nov 7. Mol Neurobiol. 2020. PMID: 31701439 Review.
Cited by
-
Moringa isothiocyanate-1 inhibits LPS-induced inflammation in mouse myoblasts and skeletal muscle.PLoS One. 2022 Dec 16;17(12):e0279370. doi: 10.1371/journal.pone.0279370. eCollection 2022. PLoS One. 2022. PMID: 36525453 Free PMC article.
-
Treatment of Periodontal Ligament Stem Cells with MOR and CBD Promotes Cell Survival and Neuronal Differentiation via the PI3K/Akt/mTOR Pathway.Int J Mol Sci. 2018 Aug 9;19(8):2341. doi: 10.3390/ijms19082341. Int J Mol Sci. 2018. PMID: 30096889 Free PMC article.
-
Glucosinolate Metabolites and Brain Health: An Updated Review on Their Potential Benefits in Neurodegenerative, Neurodevelopmental, and Psychiatric Disorders.Antioxidants (Basel). 2025 Jul 2;14(7):818. doi: 10.3390/antiox14070818. Antioxidants (Basel). 2025. PMID: 40722922 Free PMC article. Review.
-
Selected Seeds as Sources of Bioactive Compounds with Diverse Biological Activities.Nutrients. 2022 Dec 30;15(1):187. doi: 10.3390/nu15010187. Nutrients. 2022. PMID: 36615843 Free PMC article. Review.
-
Protective Effect of Glucosinolates Hydrolytic Products in Neurodegenerative Diseases (NDDs).Nutrients. 2018 May 8;10(5):580. doi: 10.3390/nu10050580. Nutrients. 2018. PMID: 29738500 Free PMC article. Review.
References
-
- Compston A, Coles A. Multiple sclerosis. Lancet 2008; 372: 1502–1517. - PubMed
-
- Siffrin V, Brandt AU, Herz J, et al. New insights into adaptive immunity in chronic neuroinflammation. Adv Immunol 2007; 96: 1–40. - PubMed
-
- Archibald CJ, McGrath PJ, Ritvo PG, et al. Pain prevalence, severity and impact in a clinic sample of multiple sclerosis patients. Pain 1994; 58: 89–93. - PubMed
-
- Osterberg A, Boivie J, Thuomas KA. Central pain in multiple sclerosis—Prevalence and clinical characteristics. Eur J Pain 2005; 9: 531–542. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

