KDIGO Clinical Practice Guideline on the Evaluation and Care of Living Kidney Donors
- PMID: 28742762
- PMCID: PMC5540357
- DOI: 10.1097/TP.0000000000001769
KDIGO Clinical Practice Guideline on the Evaluation and Care of Living Kidney Donors
Abstract
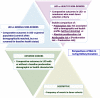
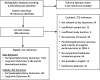
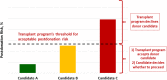
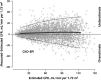
The 2017 Kidney Disease: Improving Global Outcomes (KDIGO) Clinical Practice Guideline on the Evaluation and Care of Living Kidney Donors is intended to assist medical professionals who evaluate living kidney donor candidates and provide care before, during and after donation. The guideline development process followed the Grades of Recommendation Assessment, Development, and Evaluation (GRADE) approach and guideline recommendations are based on systematic reviews of relevant studies that included critical appraisal of the quality of the evidence and the strength of recommendations. However, many recommendations, for which there was no evidence or no systematic search for evidence was undertaken by the Evidence Review Team, were issued as ungraded expert opinion recommendations. The guideline work group concluded that a comprehensive approach to risk assessment should replace decisions based on assessments of single risk factors in isolation. Original data analyses were undertaken to produce a "proof-in-concept" risk-prediction model for kidney failure to support a framework for quantitative risk assessment in the donor candidate evaluation and defensible shared decision making. This framework is grounded in the simultaneous consideration of each candidate's profile of demographic and health characteristics. The processes and framework for the donor candidate evaluation are presented, along with recommendations for optimal care before, during, and after donation. Limitations of the evidence are discussed, especially regarding the lack of definitive prospective studies and clinical outcome trials. Suggestions for future research, including the need for continued refinement of long-term risk prediction and novel approaches to estimating donation-attributable risks, are also provided.In citing this document, the following format should be used: Kidney Disease: Improving Global Outcomes (KDIGO) Living Kidney Donor Work Group. KDIGO Clinical Practice Guideline on the Evaluation and Care of Living Kidney Donors. Transplantation. 2017;101(Suppl 8S):S1-S109.
Conflict of interest statement
Kidney Disease: Improving Global Outcomes (KDIGO) makes every effort to avoid any actual or reasonably perceived conflicts of interest that may arise from an outside relationship or a personal, professional, or business interest of a member of the work group. All members of the work group are required to complete, sign, and submit a disclosure and attestation form showing all such relationships that might be perceived as or are actual conflicts of interest. This document is updated annually and information is adjusted accordingly. All reported information is published in its entirety at the end of the document and is kept on file at KDIGO.
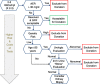
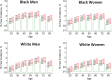
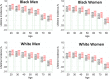
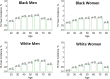
Figures




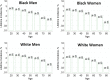
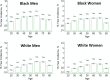
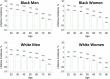
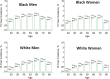
References
-
- Uhlig K, Macleod A, Craig J, et al. Grading evidence and recommendations for clinical practice guidelines in nephrology. A position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int. 2006;70:2058–2065. - PubMed
-
- Ethics Committee of the Transplantation Society. The consensus statement of the Amsterdam Forum on the Care of the Live Kidney Donor. Transplantation 2004;78:491–492. - PubMed
-
- Slinin Y, Brasure M, Eidman K, et al. Long-term outcomes of living kidney donation. Transplantation. 2016;100:1371–1386. - PubMed
-
- White CM, Ip S, McPheeters M, et al. Using existing systematic reviews to replace de novo processes in conducting comparative effectiveness reviews. In: Methods Guide for Effectiveness and Comparative Effectiveness Reviews [Internet]. Rockville (MD): Agency for Healthcare Research and Quality (US); 2008-.AHRQ Methods for Effective Health Care; 2009. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

