MAM-E17 rat model impairments on a novel continuous performance task: effects of potential cognitive enhancing drugs
- PMID: 28744563
- PMCID: PMC5591806
- DOI: 10.1007/s00213-017-4679-5
MAM-E17 rat model impairments on a novel continuous performance task: effects of potential cognitive enhancing drugs
Abstract
Rationale: Impairments in attention and inhibitory control are endophenotypic markers of neuropsychiatric disorders such as schizophrenia and represent key targets for therapeutic management. Robust preclinical models and assays sensitive to clinically relevant treatments are crucial for improving cognitive enhancement strategies.
Objectives: We assessed a rodent model with neural and behavioral features relevant to schizophrenia (gestational day 17 methylazoxymethanol acetate treatment (MAM-E17)) on a novel test of attention and executive function, and examined the impact of putative nootropic drugs.
Methods: MAM-E17 and sham control rats were trained on a novel touchscreen-based rodent continuous performance test (rCPT) designed to closely mimic the human CPT paradigm. Performance following acute, systemic treatment with an array of pharmacological compounds was investigated.
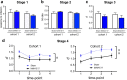
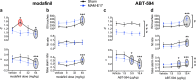
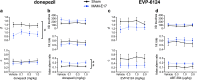
Results: Two cohorts of MAM-E17 rats were impaired on rCPT performance including deficits in sensitivity (d') and increased false alarm rates (FARs). Sulpiride (0-30 mg/kg) dose-dependently reduced elevated FAR in MAM-E17 rats whereas low-dose modafinil (8 mg/kg) only improved d' in sham controls. ABT-594 (5.9-19.4 μg/kg) and modafinil (64 mg/kg) showed expected stimulant-like effects, while LSN2463359 (5 mg/kg), RO493858 (10 mg/kg), atomoxetine (0.3-1 mg/kg), and sulpiride (30 mg/kg) showed expected suppressant effects on performance across all animals. Donepezil (0.1-1 mg/kg) showed near-significant enhancements in d', and EVP-6124 (0.3-3 mg/kg) exerted no effects in the rCPT paradigm.
Conclusion: The MAM-E17 model exhibits robust and replicable impairments in rCPT performance that resemble attention and inhibitory control deficits seen in schizophrenia. Pharmacological profiles were highly consistent with known drug effects on cognition in preclinical and clinical studies. The rCPT is a sensitive and reliable tool with high translational potential for understanding the etiology and treatment of disorders affecting attention and executive dysfunction.
Keywords: ADHD; Animal model; Attention; Behavioral pharmacology; Executive function; MAM-E17; Schizophrenia; Touch screen.
Conflict of interest statement
Conflict of interest
TWR discloses consultancy with Cambridge Cognition, Lilly, Lundbeck, and GlaxoSmithKline and has research grants with Lilly, Lundbeck, and GlaxoSmithKline. LMS and TJB consult for Campden Instruments, Ltd. The other authors declare that they have no competing interests.
Figures


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

