Defective positioning in granulomas but not lung-homing limits CD4 T-cell interactions with Mycobacterium tuberculosis-infected macrophages in rhesus macaques
- PMID: 28745326
- PMCID: PMC5785573
- DOI: 10.1038/mi.2017.60
Defective positioning in granulomas but not lung-homing limits CD4 T-cell interactions with Mycobacterium tuberculosis-infected macrophages in rhesus macaques
Abstract
Protection against Mycobacterium tuberculosis (Mtb) infection requires CD4 T cells to migrate into the lung and interact with infected macrophages. In mice, less-differentiated CXCR3+ CD4 T cells migrate into the lung and suppress growth of Mtb, whereas CX3CR1+ terminally differentiated Th1 cells accumulate in the blood vasculature and do not control pulmonary infection. Here we examine CD4 T-cell differentiation and lung homing during primary Mtb infection of rhesus macaques. Mtb-specific CD4 T cells simultaneously appeared in the airways and blood ∼21-28 days post exposure, indicating that recently primed effectors are quickly recruited into the lungs after entering circulation. Mtb-specific CD4 T cells in granulomas display a tissue-parenchymal CXCR3+CX3CR1-PD-1hiCTLA-4+ phenotype. However, most granuloma CD4 T cells are found within the outer lymphocyte cuff and few localize to the myeloid cell core containing the bacilli. Using the intravascular stain approach, we find essentially all Mtb-specific CD4 T cells in granulomas have extravasated across the vascular endothelium into the parenchyma. Therefore, it is unlikely to be that lung-homing defects introduced by terminal differentiation limit the migration of CD4 T cells into granulomas following primary Mtb infection of macaques. However, intralesional positioning defects within the granuloma may pose a major barrier to T-cell-mediated immunity during tuberculosis.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




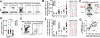


References
-
- Orme IM. The kinetics of emergence and loss of mediator T lymphocytes acquired in response to infection with Mycobacterium tuberculosis. J Immunol. 1987;138(1):293–298. - PubMed
-
- Lin PL, Rutledge T, Green AM, Bigbee M, Fuhrman C, Klein E, et al. CD4 T cell depletion exacerbates acute Mycobacterium tuberculosis while reactivation of latent infection is dependent on severity of tissue depletion in cynomolgus macaques. AIDS Res Hum Retroviruses. 2012;28(12):1693–1702. - PMC - PubMed
-
- Yao S, Huang D, Chen CY, Halliday L, Wang RC, Chen ZW. CD4+ T cells contain early extrapulmonary tuberculosis (TB) dissemination and rapid TB progression and sustain multieffector functions of CD8+ T and CD3-lymphocytes: mechanisms of CD4+ T cell immunity. J Immunol. 2014;192(5):2120–2132. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

