Buwchitin: A Ruminal Peptide with Antimicrobial Potential against Enterococcus faecalis
- PMID: 28748180
- PMCID: PMC5506224
- DOI: 10.3389/fchem.2017.00051
Buwchitin: A Ruminal Peptide with Antimicrobial Potential against Enterococcus faecalis
Abstract
Antimicrobial peptides (AMPs) are gaining popularity as alternatives for treatment of bacterial infections and recent advances in omics technologies provide new platforms for AMP discovery. We sought to determine the antibacterial activity of a novel antimicrobial peptide, buwchitin, against Enterococcus faecalis. Buwchitin was identified from a rumen bacterial metagenome library, cloned, expressed and purified. The antimicrobial activity of the recombinant peptide was assessed using a broth microdilution susceptibility assay to determine the peptide's killing kinetics against selected bacterial strains. The killing mechanism of buwchitin was investigated further by monitoring its ability to cause membrane depolarization (diSC3(5) method) and morphological changes in E. faecalis cells. Transmission electron micrographs of buwchitin treated E. faecalis cells showed intact outer membranes with blebbing, but no major damaging effects and cell morphology changes. Buwchitin had negligible cytotoxicity against defibrinated sheep erythrocytes. Although no significant membrane leakage and depolarization was observed, buwchitin at minimum inhibitory concentration (MIC) was bacteriostatic against E. faecalis cells and inhibited growth in vitro by 70% when compared to untreated cells. These findings suggest that buwchitin, a rumen derived peptide, has potential for antimicrobial activity against E. faecalis.
Keywords: Enterococcus faecalis; antibiotic resistance; antimicrobial activity; antimicrobial peptides; metagenomics; microbiome; rumen bacteria.
Figures


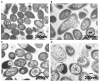
References
-
- Baddour L. M., Wilson W. R., Bayer A. S., Fowler V. G., Bolger A. F., Taubert K. A., et al. (2005). Infective endocarditis: diagnosis, antimicrobial therapy, and management of complications: a statement for healthcare professionals from the committee on rheumatic fever, endocarditis, and kawasaki disease, council on cardiovascular disease in the young, and the councils on clinical cardiology, stroke, and cardiovascular surgery and anesthesia, American Heart Association: endorsed by the infectious diseases society of America. Circulation 111, e394–e434. 10.1161/circulationaha.105.165564 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

